Global partnership leads to improved treatment for childhood diarrhea

Diarrheal diseases remain a major childhood health problem in many parts of the world. Each year, they lead to the deaths of more than 500,000 children under age 5 globally. A new large clinical trial based in Tanzania and India, led by investigators at Boston Children’s Hospital, local research universities, and the World Health Organization (WHO), finds that a revised treatment strategy of zinc supplementation improves outcomes in children with diarrhea. Results of the clinical trial were published in the New England Journal of Medicine.
Key takeaways:
- Over 500,000 children globally die each year from diarrheal diseases.
- Zinc supplementation can be an effective treatment but causes vomiting at recommended doses.
- A new clinical trial led by Boston Children’s finds a safer, effective zinc supplementation treatment strategy.
“Diarrheal diseases are an under-appreciated cause of death and morbidity among children globally,” says U.S. principal investigator of the study Christopher Duggan, MD, MPH, director of the Center for Nutrition in the Division of Gastroenterology, Hepatology and Nutrition at Boston Children’s. “While we are rightfully concerned that the current global pandemic of COVID-19 has lead to nearly 200,000 deaths in our country alone, we need to remember that diarrhea leads to more than twice as many deaths each year, all among young children,” he adds.

Many studies of childhood diarrhea in low- and middle-income countries have shown a benefit from supplemental zinc. “These studies showed that despite an increase in vomiting, supplemental zinc was associated with reduced diarrhea duration and stool output, and in some cases, reduced chances of other subsequent infections,” says Duggan. “So the downside of vomiting, which can occur because of zinc’s bitter taste, was always compared to the upside of all these other beneficial effects.” But these studies generally included very high zinc doses. Duggan’s team wanted to see if they could achieve the beneficial effects of zinc supplementation with a better safety profile.
In the recently completed trial, known as the Zinc Therapeutic Dose Trial (ZTDT), 4,500 children ages 6 months to 5 years in India and Tanzania with acute diarrhea received one of three zinc sulfate doses: 5, 10, or 20 mg daily for 14 days. The effectiveness of each dose was assessed by diarrhea duration. Safety was measured by vomiting frequency.
Similar diarrhea control with less vomiting
The results of the ZTDT showed that the proportion of children with diarrhea duration of more than five days was similar across the three treatment groups. However, the risk of vomiting was 19 percent and 29 percent lower in the 10 and 5 mg arms respectively, compared with the 20 mg arm, the dose currently recommended by the WHO.
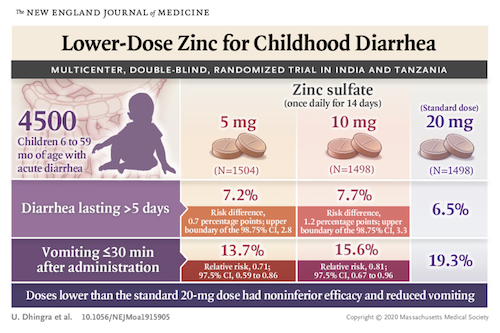
“This study shows that lower doses work equally well as higher doses but have an improved safety profile,” adds Duggan. “We think that policymaking bodies including WHO, UNICEF, and others will look at these data with great interest because one of the potential limitations in global uptake of zinc recommendations has been its association with vomiting. We are hopeful that country-based programs will promote zinc more, and that parents will be more interested in using it for their children.”
The latest research from long-term presence in Tanzania

Members of the ZTDT clinical trial in Tanzania. L-R bottom row: Research nurse Juliana Mghamba, research physician Kristina Lugangira, Professor Karim Manji (all of Muhimbili University of Health and Allied Sciences, Dar es Salaam, Tanzania); Dr. Jon Simon (World Health Organization). L-R top row: Christopher Duggan, Per Ashorn (WHO)
Duggan, who is an advisor to the Global Health Program at Boston Children’s and a Professor in the Department of Nutrition at the Harvard T.H. Chan School of Public Health, has been working in Tanzania for nearly 20 years with colleagues including Karim Manji, professor of pediatrics at Muhimbili University of Health and Allied Sciences (MUHAS) in Dar es Salaam. Over the years, Duggan, Manji, and colleagues have undertaken dozens of clinical studies on the role of nutrition in maternal and child health, with support by the National Institutes of Health, the Gates Foundation, and other sources.
Other members of the Zinc Therapeutic Dose Trial include Sunil Sazawal, Usha Dhingra, Pratibha Dhingra, Arup Dutta, Saikat Deb, Prabhabati Devi, Aishwarya Chauhan, Jitendra Kumar, and Om Prakash Semwal from the Centre for Public Health Kinetics in New Delhi, India; Karim Manji, Rodrick Kisenge, Sarah Somji, Mohamed Bakari, and Said Aboud of MUHAS; Christopher R. Sudfeld of Harvard T.H. Chan School of Public Health, Boston; Enju Liu of Boston Children’s; and Rajiv Bahl, Per Ashorn, and Jonathon Simon from WHO, Geneva, Switzerland.
Learn more about the Division of Gastroenterology, Hepatology and Nutrition and Center for Nutrition at Boston Children’s
Related Posts :
-

Team spirit: How working with an allergy psychologist got Amber back to cheering
A bubbly high schooler with lots of friends and a passion for competitive cheerleading: On the surface, Amber’s life ...
-

Thanks to Carter and his family, people are talking about spastic paraplegia
Nine-year-old Carter may be the most devoted — and popular — sports fan in his Connecticut town. “He loves all sports,” ...
-

“Observe. Be open.”: How Boston Children’s nurses are changing the future of global health
Ashley Birch, MSN, CPNP, a Boston Children’s pediatric nurse practitioner and Global Nursing fellow, didn’t expect a trash ...
-

Genetic causes of congenital diarrhea and enteropathy come into focus
Congenital diarrheas and enteropathies are rare and devastating for infants and children. Treatments have consisted mainly of fluid and nutritional ...





