Using ultrasound to trigger on-demand, site-specific pain relief
According to the CDC, 91 people die from opioid overdoses every day in the U.S. Here in Massachusetts, the state has an opioid-related death rate that is more than twice the national average.
“Opioid abuse is a growing problem in healthcare,” says Daniel Kohane, MD, PhD, a senior associate in critical care medicine at Boston Children’s and professor of anesthesiology at Harvard Medical School.
Now, Kohane and other scientists who are developing triggerable drug delivery systems at Boston Children’s Hospital have found a new way to non-invasively relieve pain without opioids. Their novel system uses ultrasound to trigger the release of nerve-blocking agents — injected into specific sites of the body ahead of time — when and where pain relief is needed most. A paper describing the findings was published online today in Nature Biomedical Engineering.
“In the future, this system could potentially combat opioid addiction by giving patients access to on-demand, non-opioid, effective local anesthesia,” says Kohane, the study’s senior investigator.
Leveraging ultrasound to treat pain
Kohane’s team has previously demonstrated that near-infrared light can be used to trigger drug release. By adapting that system to work with ultrasound, they hope the system will be even more versatile.
“One of the most interesting aspects about this system is that the degree of local anesthesia can be controlled just by adjusting the duration and intensity of the ultrasound,” says the paper’s co-first author, Alina Rwei, a graduate researcher in Kohane’s lab.
WATCH VIDEO: In 2014, Kohane gave a TED MED talk about new technologies that get drugs to where they need to go with greater precision.
Ultrasound is commercially available and widely used in various clinical and therapeutic settings, making it an attractive technology to use as a drug “trigger.” It can also penetrate deeper through tissue than light can.
“We envision that patients could get an injection at the hospital and then bring home a small, portable ultrasound device for triggering the nerve-blocking agent,” Rwei says. “This could allow patients to manage their pain relief at will, non-invasively.”
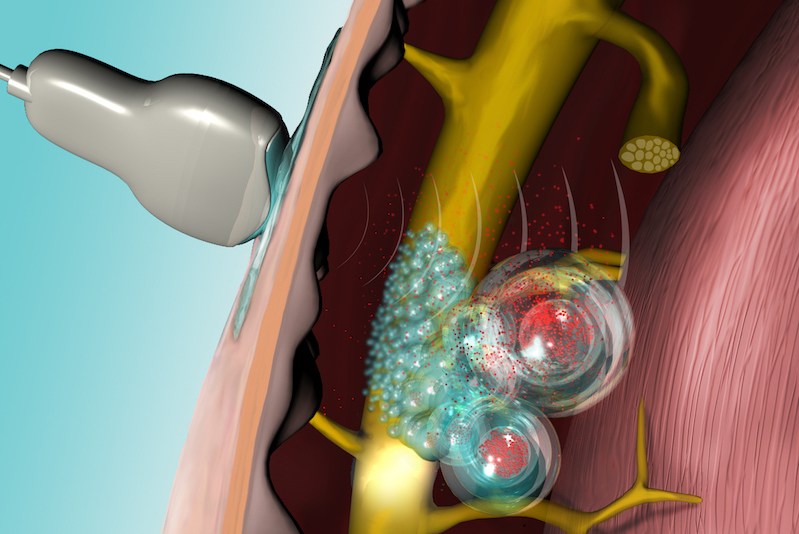
To create the system, Kohane’s team developed liposomes — artificial sacs that are micrometers in size — and filled them with a nerve-blocking drug. The walls of the liposomes contain lipids as well as small molecules called sono-sensitizers, which are sensitive to ultrasound.
Reactive oxygen species are natural byproducts of metabolizing oxygen. Here, they chemically react with and disrupt the surface of the liposomes.
“Once the drug-filled liposomes are injected, ultrasound can be applied to penetrate tissue and cause the sensitizers to create reactive oxygen species, which react with lipids in the walls of the liposomes,” Kohane says. “This opens the surface of the liposomes and releases the local anesthetic drug into the surrounding tissue or onto an adjacent nerve, reducing pain.”
Tailoring a sono-sensitive drug delivery system
The small sono-sensitizer molecules that the team built into the liposomes are the active component of a drug that is already approved by the FDA and currently used in photodynamic therapy. Right now, the Kohane lab’s system can be activated by ultrasound for up to three days after the liposomes are injected, positioning it well for future translation as a strategy to manage post-operative pain.
“Out of all the particle delivery systems, I think liposomes are one of the most clinically accepted and customizable options out there,” Rwei says. “Our research indicates that liposomes can be tailored to respond to near-infrared light, ultrasound and even magnetic triggers.”
In addition to Kohane and Rwei, the paper’s other contributors are co-first author Juan L. Paris, and co-authors Bruce Wang, Weiping Wang, Christopher D. Axon, Maria Vallet-Regi and Robert Langer.
This work was supported by the National Institutes of Health (GM073626) and the Ministerio de Economía y Competitividad, Spain (BES-2013-064182, EEBB- I-16-11313 associated with MAT2012-35556).
Learn more about drug delivery research at Boston Children’s
Related Posts :
-

Nanoparticle drug combo treats venous malformations
Venous malformations are abnormally shaped veins that develop when the cells lining the blood vessels grow too fast when they ...
-

The journey to a treatment for hereditary spastic paraplegia
In 2016, Darius Ebrahimi-Fakhari, MD, PhD, then a neurology fellow at Boston Children’s Hospital, met two little girls with spasticity ...
-

A safe, pain-specific anesthetic shows preclinical promise
All current local anesthetics block sensory signals — pain — but they also interrupt motor signals, which can be problematic. For example, ...
-

Which pain medication is right for your child? What a pediatrician wants parents to know
There’s no shortage of safe and effective pain medications for children. Acetaminophen (commonly known as Tylenol), ibuprofen (Motrin, Advil), ...





