Novel CRISPR system could halt growth of triple-negative breast cancer

Triple-negative breast cancer (TNBC), lacking estrogen, progesterone and HER2 receptors, has the highest mortality rate of all breast cancers. It more frequently strikes women under age 50, African American women, and women carrying a BRCA1 gene mutation. The highly aggressive, frequently metastatic cancer is in urgent need of more effective targeted therapeutics.
A new tumor-targeted CRISPR gene editing system, encapsulated in a nanogel and injected into the body, could offer a genetic treatment, suggest researchers at Boston Children’s Hospital. In a proof-of-principle study, conducted in human tumor cells and live, tumor-bearing mice, the CRISPR system effectively halted the growth of TNBC while sparing normal cells. Peng Guo, PhD, Marsha Moses, PhD and their colleagues report the findings today in PNAS.
Precision delivery of CRISPR
To date, a lack of effective delivery systems has limited the translation of CRISPR gene editing into therapies. One method uses a virus to deliver CRISPR, but the virus cannot carry large payloads and potentially can cause side effects if it “infects” cells other than those targeted. Another method packages the CRISPR tools inside a cationic polymer or lipid nanoparticles. But these elements can be toxic to cells, and the body often traps or breaks down the nanoparticles before they reach their destination.
The new approach encapsulates the CRISPR editing system inside a soft “nanolipogel” made up of a nontoxic double layer of fatty molecules and a hydrogel. Antibodies attached to the gel’s surface then guide the CRISPR nanoparticles to the tumor site. (The antibodies are designed to recognize and target ICAM-1, a therapeutic target for TNBC discovered by the Moses Lab in 2014.)
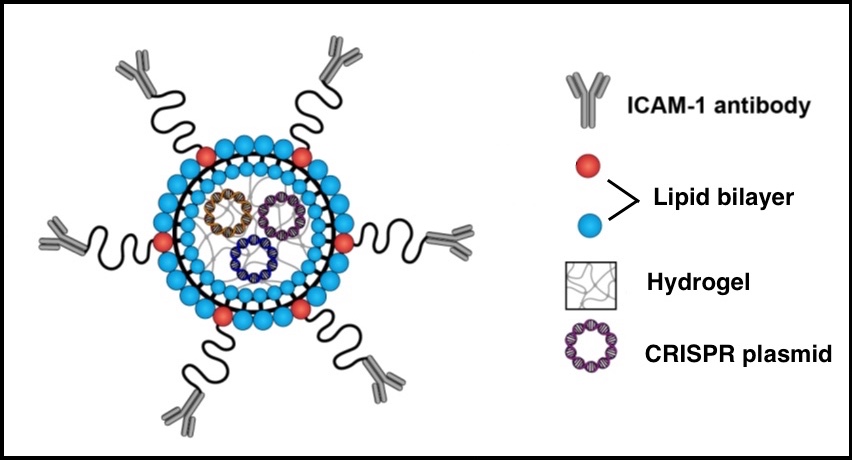
Because the particles are soft and flexible, they can enter cells more efficiently than their stiffer counterparts. Stiffer nanoparticles tend to get trapped by the cell’s ingestion machinery, while the soft particles fused with the tumor cell membrane and delivered their CRISPR payloads directly inside the cell, the researchers found.
“Using a soft particle allows us to penetrate the tumor better, without side effects, and with bigger cargo,” says Guo, the study’s first author. “Our system can substantially increase tumor delivery of CRISPR.”
Knocking out a breast cancer oncogene
Once inside the cell, the CRISPR system knocked out lipocalin 2, an oncogene that promotes breast tumor progression and metastasis, with an editing efficiency of 81 percent in tumor tissue. Experiments showed that loss of the oncogene inhibited the cancer’s aggressiveness and tendency to migrate or metastasize. Overall, the approach attenuated tumor growth by 77 percent in live, tumor-bearing mice and showed no toxicity in normal tissues.
Although the study focused on triple-negative breast cancer, Moses, who directs Boston Children’s Vascular Biology Program, believes the team’s CRISPR platform could be adapted to treat pediatric cancers as well, and could also deliver conventional drugs. These studies are ongoing. The team is in discussions with a number of companies interested in the technology.
“Our system can deliver significantly more drug to the tumor, in a precise and safe way,” Moses says.
The study was funded by the National Institutes of Health and the Breast Cancer Research Foundation. Guo, Moses and coauthors Jiang Yang, PhD (Boston Children’s Hospital) and Debra Auguste, PhD (Northeastern University) are co-inventors on a patent application filed by Boston Children’s Hospital. Coauthor Jing Huang, PhD, is a consultant for Simcere Pharmaceutical Company. The other authors declare no other competing interests.
To inquire about the technology, email Adrienne.Wong@childrens.harvard.edu.
More stories from the Vascular Biology Program here.
Related Posts :
-

Mapping ‘neighborhoods’ in aggressive childhood brain tumors
The third-most common kind of childhood brain tumor, supratentorial ependymoma, is an aggressive cancer that often returns after surgery and ...
-

Gene therapy for hearing loss: Tag-teaming from the lab to the clinic
Two-year-old Miles is one of the first patients with hereditary hearing loss to receive gene therapy at Boston Children’s ...
-

A true hero’s journey: How a team approach helped Wolfie overcome pancreatitis
Wolfgang, affectionately known as “Wolfie,” is a bright and energetic 7-year-old with a quick wit and a love for making ...
-

The journey to a treatment for hereditary spastic paraplegia
In 2016, Darius Ebrahimi-Fakhari, MD, PhD, then a neurology fellow at Boston Children’s Hospital, met two little girls with spasticity ...





