Reconstructing a chest wall, one virtual step at a time
It takes a village of clinicians and engineers to reconstruct a chest wall. It also takes a lot of 3D modeling.
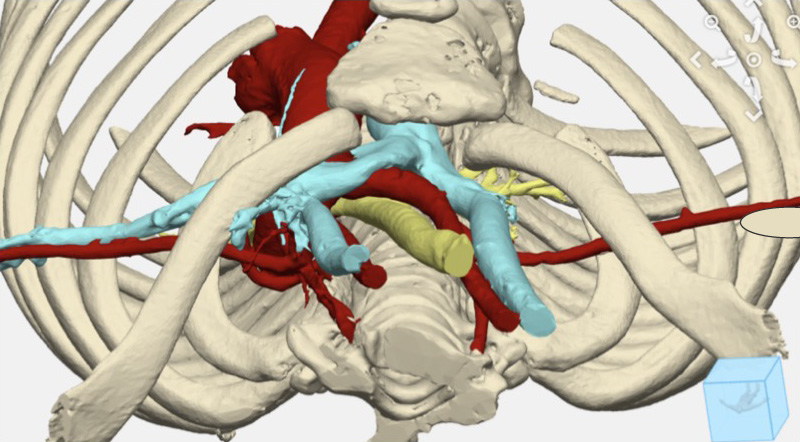
A young girl needed to have her chest wall restructured to stop life-threatening airway obstruction. To ensure the complicated operation would be efficient, Emily Eickhoff, a Boston Children’s biomedical engineer, partnered with a team of surgeons from various disciplines to create 3D models that represented the anatomy of the patient’s chest. Those detailed viewpoints gave the surgeons an informed head start before they walked into the operating room: They could visualize how to precisely divide and enlarge her sternum and study how implanted titanium plates would support an expansion of her chest wall. That expansion would create enough room to improve her airway.
“It was amazing to be a part of the solution — a child who was having trouble breathing is now able to breathe,” says Eickhoff.
Innovating to simplify the complex
Christopher Baird, MD, lead cardiac surgeon of the Vascular Ring and Airway Compression Program, had long wanted to solve a puzzle that made chest wall reconstruction challenging. A surgeon has to know in advance the exact amount of space needed in a patient’s chest wall to avoid compression of the airway, aortic vessels, and esophagus. Baird wondered if 3D modeling could create a clear and accurate roadmap for how to approach the operation.
“For this model, we had to take apart the chest anatomy and look at it in all different ways.” — Cardiac surgeon David Hoganson
Baird turned to fellow cardiac surgeon David Hoganson, MD, who oversees a computational 3D visualization initiative at the Benderson Family Heart Center. “There were a lot of similarities in preparing a chest wall reconstruction to how we already make patient-specific 3D models for the preoperative planning of other complex cases,” Hoganson says. “For this model, we had to take apart the chest anatomy and look at it in all different ways.”
The top opening of the girl’s chest cavity was narrow. The opening is known as the thoracic inlet, and in her case, the narrowing caused her airway to be compressed between her aorta and spine. She had already experienced cardiac arrest because of the condition, so it was imperative to quickly correct it.
Digital modeling leads to tangible results
Over a couple of weeks, Eickhoff created 3D digital models of the patient’s chest based on CT scans. “It was a new challenge, which was invigorating,” she says. “I had to understand the relationship between the ribs and the spine, and how that defines the front-to-back dimension of the chest.”
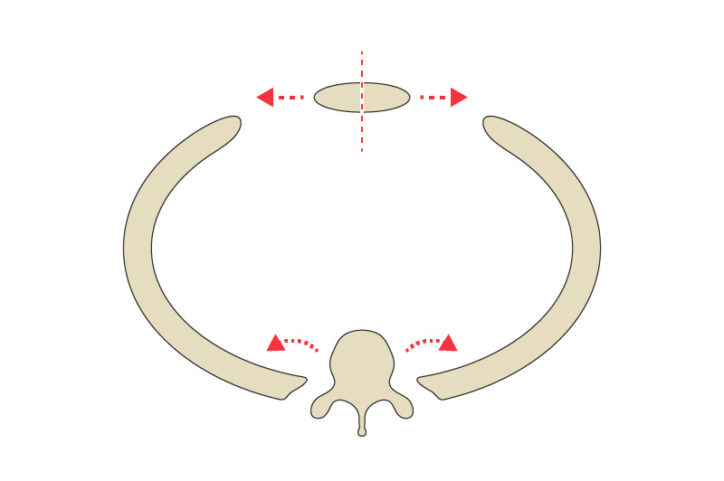
The girl had two centimeters of space between her sternum, in the front, and her spine in the back — not much room to accommodate the critical components in that area, including vessels that carry blood to the brain. Eickhoff, consulting with clinicians as she worked, created several models to simulate how repositioning the ribs and sternum would affect the width of the thoracic inlet. By performing these virtual operations, the surgeons could see how different decisions might affect the chest wall anatomy.
The team ultimately decided to expand the sternum with small sections of rib, which enlarges the thoracic inlet. The digital model created from that plan was used to make a life-sized 3D-printed model so they could get a tangible sense of the reconstructed chest wall. It also allowed the surgeons to determine the proper size and shape of the titanium plates that would stabilize the chest wall.
Specialists from across Boston Children’s had unique roles
The final step was an all-day operation that involved surgeons and specialists from eight subspecialities. “It was a multi-team approach that used everyone’s individual skills and knowledge,” says Brian Snyder, MD, PhD, an orthopedic surgeon.
According to Daniel M. Balkin, MD, PhD, a plastic surgeon, “The sternum was split and her airway and aortic vessels were mobilized and repositioned by cardiac and general surgeons. The chest wall was stabilized in its expanded form using bone graft and titanium plates by orthopedic surgeons. And plastic surgeons covered the entire construct with muscle flaps for protection and to optimize healing.”
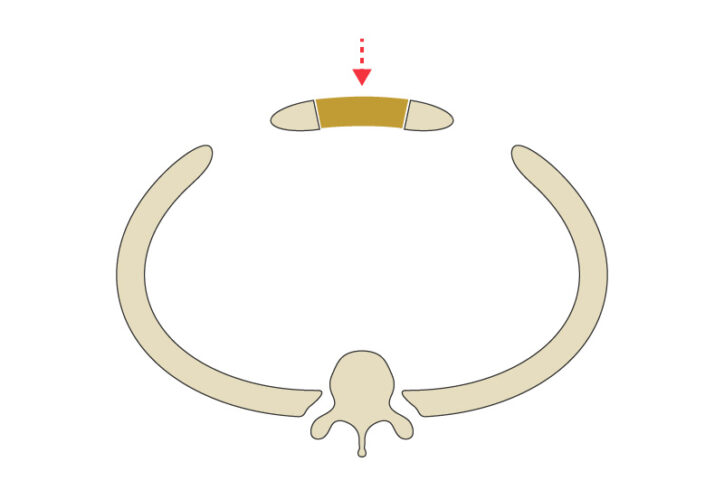
The team expanded the girl’s chest wall from 22 millimeters to 35.5 millimeters. That created the necessary space to improve her airway through a surgical procedure known as a tracheopexy, adds Benjamin Zendejas-Mummert, MD, MSc, an esophageal and airway surgeon. “We were able to offload pressure from her trachea.”
Boston Children’s cardiologists, otolaryngologists, and pulmonologists also participated in the operation — one that started weeks earlier on a computer screen.
“It was a multi-team approach that used everyone’s individual skills and knowledge.” — Orthopedic surgeon Brian Snyder
Chasing perfection to improve children’s health
“The rewarding part of this is helping a child be able to breathe without difficulty,” Baird says. “She and others we treat can enjoy active lives.”
Since operating on the girl, Baird and colleagues have relied on 3D modeling for other chest wall reconstructions. And with a foundation now set, Eickhoff needs only a matter of days to prepare a new digital model.
“As engineers, we’re chasing perfection,” Hoganson says. “So, it’s great when these moments happen, when we take all these innovative capabilities and make a difference.”
Learn more about the Vascular Ring and Airway Compression Program and the Esophageal and Airway Treatment Center, or schedule a second opinion.
Related Posts :
-

After vascular ring surgery, playing and eating are a breeze for Louis
You can’t blame 10-year-old Louis McFaye for eating three hot dogs really fast. Until last summer, he struggled to ...
-

'They just cared about Levi': Finding answers for a complex vascular ring
The first sign that Levi Blair Winkler would no longer have problems swallowing was when, only three days after a ...
-

A heart valve that grows along with a child could reduce invasive surgeries
Clinical trials have started for the first prosthetic pulmonary valve replacement that is specifically designed for pediatric patients and can ...
-

Research 2022: Tackling disease in new ways
Researchers across Boston Children’s spent 2022 imagining new solutions to old challenges in health and medicine, opening the door to ...





