A deeper understanding of inflammatory pain could reveal new solutions
Non-steroidal anti-inflammatory drugs (NSAIDs) such as ibuprofen are the main go-to for inflammatory pain caused by wounds, infection, sunburn, arthritis, and other triggers. NSAIDs work pretty well, but chronic use can cause side effects, and they aren’t equally effective for all sources of pain. Could we identify a more effective, safer analgesic that doesn’t involve opioids?
New research from the lab of Clifford Woolf, MB, BCh, PhD, director of the F.M. Kirby Neurobiology Center at Boston Children’s Hospital, took the most fine-grained look at inflammatory pain to date. Led by Aakanksha Jain, PhD, and in collaboration with Harvard Medical School’s Laboratory of Systems Pharmacology, the team identified thousands of molecular interactions between pain-triggering neurons (nociceptors) and different types of immune cells.
These interactions, most not previously known, could produce pain hypersensitivity during inflammation — and also resolve it. Published as the cover article in Nature Immunology, the findings provide a platform to advance our understanding of pain and find better treatments.
Neuroimmune interactions
Jain and her colleagues used single-cell RNA sequencing to explore the crosstalk between nociceptors and different types of immune cells in the skin when mice were exposed to three different types of pain triggers (infection, incision, and burn). Each trigger produced extensive changes in gene expression in the different immune cell types, especially macrophages. Some changes happened right away while others took days to appear.
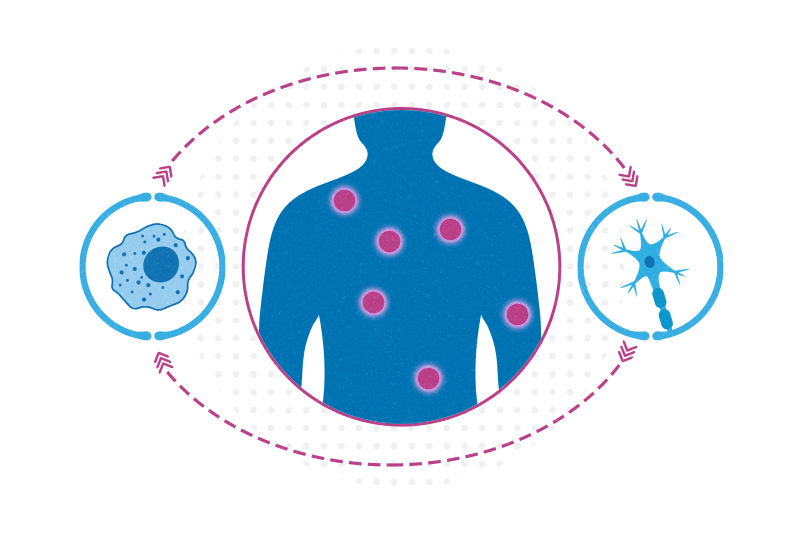
Next, based on a database of neuroimmune interactions generated through a platform called INDRA, Jain and colleagues mapped out separate cellular “interactomes” for each injury type before, during, and after a pain episode. Cell signaling went in both directions: Immune cells acted on nociceptors and vice versa.
“In the past, people looked at the nervous system’s role in pain and immune system’s role in pain,” says Woolf. “We’re saying that’s an artificial separation; the immune and nervous system work together. We have to embrace complexity.”
The key finding was that inflammatory pain isn’t a single entity. Each of the three inflammatory pain sources the researchers tested produced distinct networks of interactions between nociceptors and immune cells.
“All three pain settings have very different mechanisms driving pain,” says Jain. “Now that we have the network of interactions, we can look for ways to promote resolution of pain.”
A new analgesic for inflammatory pain?
In addition to mechanisms driving pain, the study found a factor secreted by immune cells, thrombospondin1 (TSP1), that was upregulated in all three pain models. When the team treated nociceptors with TSP1, their activity was suppressed.
“We set out to identify immune molecules that drive pain, thinking we could block them, but TSP1 kept coming up,” says Jain. “Thrombospondin had never been studied in pain or in the peripheral nervous system before.”
The researchers think that TSP1 may help restore balance, easing pain when it’s no longer needed as an alert system.
“We know there are endogenous opioids, but it was unexpected that there was something else with a pain suppressive action,” says Woolf.
In the future, the researchers plan further studies of TSP1 and the CD47 receptor it acts on to see if they can develop a pain treatment. They also plan to use their discovery platform to find new factors that cause or help resolve pain. Woolf sees applications in treating pain from trauma and in postoperative pain management.
Ultimately, Jain hopes to go beyond the skin model used in this study to explore joint pain in rheumatoid arthritis and osteoarthritis. As Woolf puts it, “It’s literally just the beginning.”
Explore research in the F.M. Kirby Neurobiology Center.
Related Posts :
-

A bio-inspired approach to delivering local anesthetics
Site 1 sodium channel blockers such as tetrodotoxin and saxitoxin are small-molecule drugs with powerful local anesthetic properties. They provide pain ...
-

As COVID-19 fuels opioid deaths, researchers look to create an anti-opioid vaccine
A project that began one year ago at Boston Children’s Hospital to develop an anti-opioid vaccine is starting to ...
-

Hip pain is different in female dancers: New insights from dynamic ultrasound
Dancers put unique demands on their hips, achieving extreme ranges of motion that can strain the joints and damage supporting ...
-

Nerve block and a way out of pain after surgery
If she hadn’t dislocated her knee severely when she was 13, Paige Thornton probably wouldn’t have such strong feelings ...