How the antidepressant ketamine rapidly awakens the brain, and why its effects vary more in women

In small doses, the anesthetic ketamine is a mildly hallucinogenic party drug known as “Special K.” In even smaller doses, ketamine relieves depression — abruptly and sometimes dramatically, steering some people away from suicidal thoughts. Studies indicate that ketamine works in 60 to 70 percent of people not helped by slower-acting SSRIs, the usual drugs for depression.
Two ketamine-like drugs are in the clinical pipeline, and, as of this week, one appears close to FDA approval. With no significant new antidepressant in more than 30 years, anticipation is high. Yet no one has pinned down how low-dose ketamine works. Studies have implicated various brain neurotransmitters and their receptors — serotonin, dopamine, glutamate, GABA receptors, opioid receptors — but findings have been contradictory.
“We felt it was time to figure this out once and for all,” says neuroscientist Takao Hensch, PhD.
In a study published late last month in Molecular Psychiatry, the laboratories of Hensch and Michela Fagiolini, PhD, both in the F.M. Kirby Neurobiology Center at Boston Children’s Hospital, pinpointed ketamine’s mechanism of action. And, unexpectedly, they found evidence that its efficacy in females varies with the menstrual cycle — suggesting the need to time treatment accordingly.
Parsing ketamine’s action
It has widely been held that ketamine relieves depression by blocking NMDA, a glutamate receptor. Glutamate is an excitatory neurotransmitter, making neurons more active, more communicative and more likely to form new connections. So logic would suggest that blocking its receptor, depriving neurons of excitatory signals, would make the brain even more sluggish and indifferent to stimulation — worsening depression.
“Ketamine, by blocking the NMDA receptor, should dampen neural activity,” says Hensch. “But the paradox is that it makes neural activity stronger — it awakens the brain.”
The research team, led by postdoctoral fellows Nathalie Picard, PhD, and Anne Takesian, PhD, teased out the puzzle in mice using a genetic approach. They deleted the gene for one component of the NMDA receptor, called the 2A subunit. And they deleted it only in a certain kind of neuron in the brain, the parvalbumin interneurons. Then they gave the mice ketamine.
Awakening the brain
The investigators first measured the firing of neurons in the cerebral cortex, and found that it was rapidly enhanced by ketamine — except in mice whose NMDA receptors were deleted. In a swim test, the classic model for measuring antidepressant activity, mice given ketamine worked more vigorously than untreated mice to rescue themselves from the cold water — but again, only when their NMDA receptors were intact.
“With ketamine, the neurons became more active,” says Hensch. “But they were not rescued if the 2A subunit was deleted. We think this is the best evidence yet that it’s an NMDA receptor on a particular kind of cell that’s important.”
That cell type explains the paradox of ketamine’s action. Small in number and size, parvalbumin neurons synchronize the electrical activities of other brain cells. They are inhibitory in nature, depressing the activity of the brain circuits around them. Blocking glutamate eliminates this natural inhibition, exciting the circuits as a whole. This, Hensch believes, allows the brain to be more sensitive and responsive to sensory input, lifting it out of its depressive funk.
Sex-specific effects
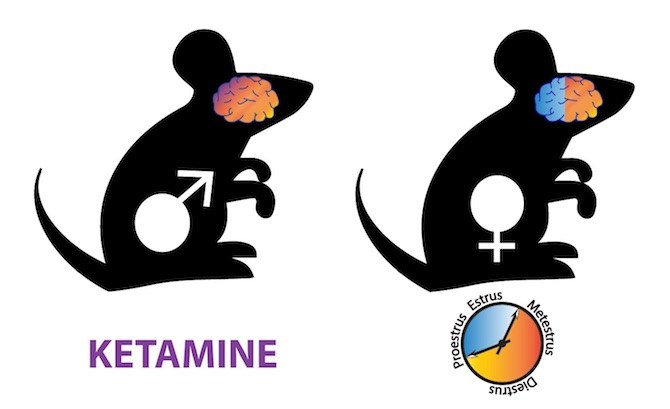
The second part of the study was more surprising: Female mice responded less consistently to ketamine.
“We found that drug action was variable – sometimes ketamine would boost activity, sometimes not,” says Hensch.
When Picard and Takesian looked at females during different stages of the estrous cycle, they found that ketamine had little effect during the follicular phase (which in women starts on the first day of menstruation and ends with ovulation). During this time, parvalbumin neurons had fewer glutamate receptors bearing the 2A subunit.
Hensch and Fagiolini think clinical trials of ketamine should take this into account. “In the clinical literature, ketamine effectiveness varies widely — from 40 to 80 percent,” says Fagiolini. “The timing of treatment in women may explain that variability.”
Insights from Rett syndrome
Hensch and Fagiolini took cues from their prior work on Rett syndrome, a rare neurologic condition in girls that causes vision loss, autism and other complications. In 2012, they showed in mice that the Rett mutation disrupts brain wiring early in development, such that parvalbumin connections were too abundant, forming many more synapses than normal. The end result was an excess of inhibition such that brain activity died down.
“The parvalbumin cells were maturing faster than normal,” says Fagiolini. “Through electrical recordings, we showed that the brain is quite normal early on, but activity is gradually lost — consistent with the ‘silencing’ of the brain that we observe in Rett syndrome.”
But when Hensch and Fagiolini deleted the NMDA receptor and, in particular, the 2A subunit, in the brains of mice with Rett syndrome, they slowed down parvalbumin-cell maturation. This prevented vision loss in the mice, mirroring reduced disease severity.
Follow-up work in Fagiolini’s lab showed that low-dose ketamine, given daily, restored normal brain circuitry, slowed the progression of Rett symptoms and extended the animals’ life span. This insight, among others, prompted a multicenter clinical trial of ketamine for Rett syndrome in girls aged 6-12, which opened in 2018 at Boston Children’s and six other centers.
The new work suggests that trials in women and older girls with Rett syndrome, an X-linked disorder, should be mindful of the menstrual cycle and when it begins in younger patients.
Cleaning up ketamine’s action
Ketamine is a “dirty” drug that acts on many receptors with many side effects, and is thought to be addictive. But the researchers think it may be possible to find a cleaner, safer antidepressant that acts through the NMDA receptor, and specifically through the 2A subunit.
Hensch’s lab is also interested in exploring the potential usefulness of modulating parvalbumin circuits in psychosis and other conditions in which these cells are altered. Abnormalities in these cells are believed to play a pivotal role in autism, schizophrenia and other neurodevelopmental disorders.
“We’re interested in how these cells develop faster or slower in different diseases,” says Hensch.
The study was funded by the Silvio Conte Center of the National Institute of Mental Health (P50MH094271), the University of Tokyo International Research Center for Neurointelligence (IRCN), the Rett Syndrome Research Trust, Rett syndrome.org, the National Institute of Neurologic Disorders and Stroke (R01 NS095959), the National Eye Institute (R01 EY013613), Pfizer, the Nancy Lurie Marks Family Foundation and the Canadian Institute for Advanced Research.
Related Posts :
-

Beyond average-based medicine: HIE as a blueprint for data-informed care
Historically, outcome prediction in medicine has followed a familiar formula: run a clinical trial, publish the results, guide care based ...
-

The hidden burden of solitude: How social withdrawal influences the adolescent brain
Adolescence is a period of social reorientation: a shift from a world centered on parents and family to one shaped ...
-

The journey to a treatment for hereditary spastic paraplegia
In 2016, Darius Ebrahimi-Fakhari, MD, PhD, then a neurology fellow at Boston Children’s Hospital, met two little girls with spasticity ...
-

Help your child manage anxiety about school violence
With news of school shootings and other violence often reaching children, parents sometimes grapple with how to help their child ...





