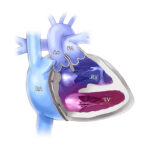
Someday, this prosthetic heart valve might be the only one a child needs
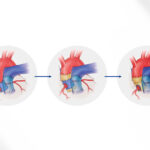
More than 330,000 children worldwide are born with a heart valve defect, and millions of others develop rheumatic heart disease requiring early valve replacement. Current prosthetic heart valves are fixed in size, so typically need to be replaced every few years as a child grows. For children receiving their first replacement before age 2, that means as many as five high-risk open-heart operations before they reach adulthood.
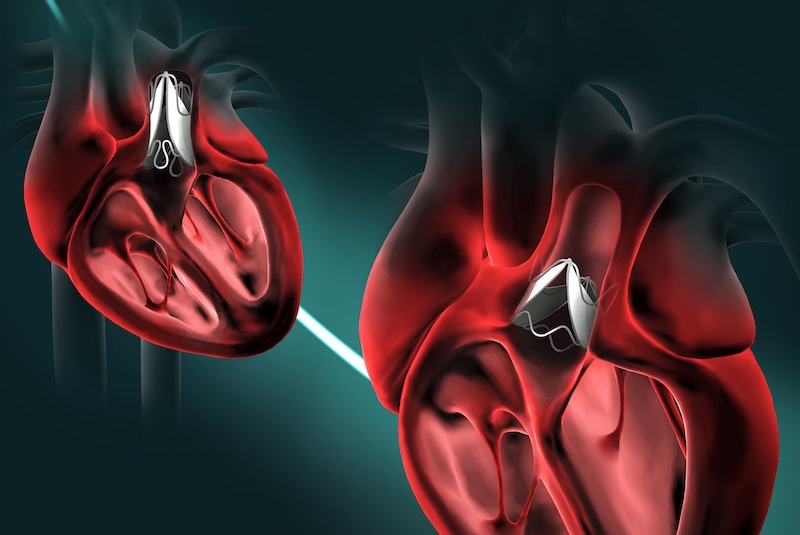
But a surprising new design created at Boston Children’s Hospital could allow children to keep the same prosthetic valve until adulthood. The research team, led by Pedro J. del Nido, MD, chairman of Cardiovascular Surgery at Boston Children’s Hospital, envision patients having the valve expanded through a minimally invasive balloon catheter procedure as needed.
As described yesterday in Science Translational Medicine, the valve underwent extensive benchtop studies, computer simulations, and testing in large animal models. All showed that the new design enables the valve to work across wide range of sizes, retaining its functionality.
“We hope to bring this new device into clinical testing fairly rapidly,” says del Nido, the paper’s senior author. “If our preclinical results hold up in human testing, this could transform the field.”
Less is more: A bileaflet heart valve
Commercially available prosthetic heart valves have three leaflets, tiny flaps that provide a one-way inlet or outlet for blood to keep it flowing in the right direction. The new design was inspired by human venous valves, found in the deep veins of the leg. Unlike our hearts’ native outflow valves, venous valves have just two leaflets. Nature has optimized their geometry to maintain closure and one-way blood flow even when the veins expand in diameter.
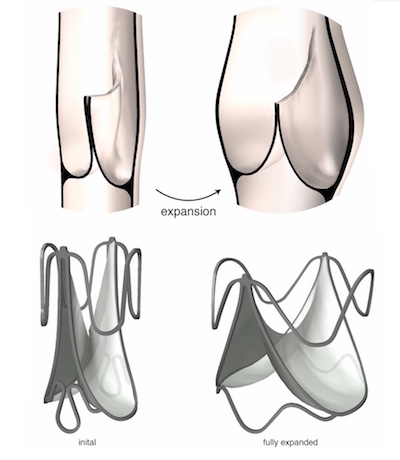
“Veins carry approximately 70 percent of our blood volume, and their dimensions can change dramatically depending on body position,” explains Sophie Hofferberth, MD, a surgical resident at Brigham and Women’s Hospital who led the research in del Nido’s Boston Children’s lab. “We mimicked the geometric profile of the human venous valve to design a bileaflet heart valve of programmed dimensions that is adaptable to growth without loss of one-way flow control.”
In multiple rounds of testing, valve prototypes were able to expand to accommodate growth and structural asymmetries within the heart. They remained fully functional across a range of dimensions, pressures, and flow rates.
Because the valve can expand without requiring the frame and leaflet to stretch or enlarge, it is compatible with a range of off-the-shelf materials, the researchers say. The researchers successfully and safely expanded the device at multiple timepoints in a growing sheep model, using a balloon catheter.
Potential for fewer blood clots
The researchers also observed that the bileaflet heart valve design encourages good blood flow through the valve. This could potentially reduce the risk for blood clots, a complication often seen with existing prosthetic valves. The team saw no evidence of blood clot formation in the growing sheep model over 10 weeks of observation, even without the use of blood-thinning medication typically given to prosthetic valve recipients.
If our preclinical results hold up in human testing, this could transform the field.
“Flow disruptions that lead to blood clot formation and early valve deterioration are a shortcoming of many existing devices,” says Hofferberth, who is first author on the paper. “Our design achieves a favorable flow profile that seems to facilitate effective valve washout and minimize flow stagnation, which is likely to be an important determinant of long-term device durability.”
The research team believes their data support initiation of a clinical study within one to two years.
More cardiac research from Boston Children’s Hospital
Mossab Saeed, Christopher Payne, Karl Price, and Peter Hammer of Boston Children’s Department of Cardiac Surgery were coauthors on the paper, together with Lara Tomholt, Matheus Fernandes, and James Weaver of the Wyss Institute for Biologically Inspired Engineering; Gerald Marx, Jesse Esch, and David Brown of Boston Children’s Department of Cardiology; Jonathan Brown and Elazer Edelman of MIT; and Richard W. Bianco of the University of Minnesota. The study was supported by a NIH-NRSA postdoctoral fellowship grant (1F32HL138993-01), an Early Career Award from the Thrasher Research Fund, and the Oakwood Foundation. A provisional patent has been filed naming several of the authors.
Related Posts :
-

Biventricular repair linked to better outcomes in superior-inferior ventricles treatment
The complexity of superior-inferior ventricles — a condition in which one ventricle is positioned on top of the other — has often ...
-

A reunion of the heart: Grace and Amy bond through HLHS
At first glance, Grace and Amy have a pretty typical friendship: they joke around, cheer each other on, and keep ...
-

First-of-its-kind pressurization test could improve Ross procedure outcomes
The Ross procedure is a preferred surgery to treat severe aortic valve disease. The procedure replaces the failing valve ...
-

Four things you should know about MAPCAs treatment
As the first grandchild in her family, Hannah Homan is in demand for frequent visits. She was also the focus ...