Building a body, one organ chip at a time
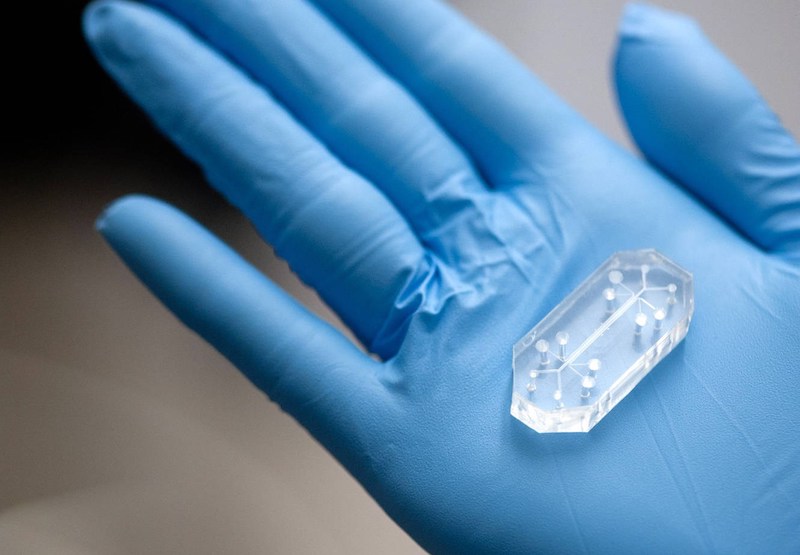
They don’t look like much sitting in your hand. A few pieces of clear plastic, each smaller than an Altoids tin, with channels visible inside and holes for plugging tubing into them.
But fill them with cells and treat those cells the right way, and they turn into something amazing: tiny hearts, lungs, guts, kidneys.
They’re “organs on chips,” and they represent what’s probably the most comprehensive effort to date to physically model the functions of whole organs for drug development and disease research.
Developed by a team of biologists and engineers led by Donald Ingber, MD, PhD, a member of Boston Children’s Hospital’s Vascular Biology Program and director of the Wyss Institute for Biologically Inspired Engineering at Harvard, they’re the building blocks for an ambitious project to create an artificial multi-organ system—essentially, a whole body on a chip.
Each of the chips—Ingber’s team is currently developing 10 different organs—is built using microfabrication techniques like those common in the semiconductor industry. “This allows us to create features and structures that we can control at the size scale in which cells live, and also apply physiological fluid flows and mechanical forces,” Ingber explains. “We have precise control over where cells live in the device and what they experience.”
Going through the motions
The two features Ingber mentioned, flow and force, are instrumental in faithfully mimicking organ function. His lung on a chip, for instance, has a central microfluidic channel that is split into two parallel channels by porous flexible membrane. One side of the membrane is coated with human lung “air sac” cells (over which air can pass), the other with human lung capillary blood vessel cells. A combination of pumps subject the cells to the sensations of continuous blood flow and rhythmic breathing by moving culture medium through the blood vessel channel and applying suction that deforms the cell-coated membrane.
This video from the Wyss Institute explains in more detail:
Those forces incite dramatic responses from the cells in the chips. “You can put endothelial [blood vessel] and epithelial [air sac] cell cultures together, but they won’t reflect the range of functions you see in a real lung,” Ingber says. “This is because respiratory physiology relies on the mechanics of breathing and blood flow.
“For instance, once we added breathing motions to the chip,” he adds, “the epithelial cells started producing surfactant, just like what happens in the lining of a normal lung when a baby takes its first breath.”
“[O]nce we added breathing motions to the [lung] chip, the epithelial cells started producing surfactant, just like what happens…when a baby takes its first breath.”
Similarly, Ingber’s lab recently revealed that breathing motions were essential for using the lung chip to mimic the pulmonary edema (fluid leakage from blood vessels into the lungs) that occurs in some cancer patients treated with the drug interleukin-2—the first demonstration that chips could model a complex human disease, as well as a drug toxicity.
Ingber’s team has also developed a chip that models the complex environment of the human gut—including its microbial inhabitants. “Once we added a trickling flow of medium and peristaltic motions, the cells in the chip started forming structures similar to intestinal villi,” he says. “Now we’ve added bacteria, and can start to study the relationship between the microbiome and human intestinal disease processes.”

With a host of chips now in hand, Ingber and his team are now working with the federal Defense Advanced Research Project Agency to create a system for linking chips representing different organs together. The system will allow broader study of organ physiology and also how drugs affect multiple organs—the first step toward simulating a complete living body.
“We want to be able to administer a drug via the ‘gut’ or ‘lungs,’ see how it is metabolized by the ‘liver,’ excreted by the ‘kidney,’ and whether it causes toxicity in the ‘heart,’” says Ingber. “We also want to model a broad range of disease states, like asthma, Crohn’s, radiation exposure and so on.”
He’s also talking to the Food and Drug Administration about potentially accepting organ-on-chip data as part of the drug approval process in addition to or, in the future, in lieu of animal data. “We’ve only just started the discussion, but we hope they’ll one day accept human organ chip data instead of certain animal studies, just as they’ve begun to accept biomarker data.”
Related Posts :
-

Four things you should know about MAPCAs treatment
As the first grandchild in her family, Hannah Homan is in demand for frequent visits. She was also the focus ...
-

Treating MAPCAs with unifocalization surgery and cardiology care
Children born with a rare form of tetralogy of Fallot (ToF) face a challenging type of congenital heart ...
-

The journey to a treatment for hereditary spastic paraplegia
In 2016, Darius Ebrahimi-Fakhari, MD, PhD, then a neurology fellow at Boston Children’s Hospital, met two little girls with spasticity ...
-

A unique marker for pericytes could help forge a new path for pulmonary hypertension care
Pulmonary arterial hypertension (PAH) is a rare condition that’s difficult to treat. The hallmarks of the disease — narrowing of ...





