Targeting a rogue T cell prevents and reverses multiple sclerosis in mice
Multiple sclerosis is an autoimmune disease affecting both adults and children. It’s driven by “helper” T cells, white blood cells that mount an inflammatory attack on the brain and spinal cord, degrading the protective myelin sheath that covers nerve fibers. But there are many different kinds of T helper cells, and up until now, no one knew which ones were the bad actors.
Researchers at Boston Children’s Hospital have now pinpointed the specific helper T cells that cause MS, as well as a protein on their surface that marks them. As reported this week in PNAS, an antibody targeting this protein, CXCR6, both prevented and reversed MS in a mouse model.
If human studies bear out the findings, targeting these rogue T cells could potentially ameliorate MS, says senior investigator Eileen Remold-O’Donnell, PhD, of the hospital’s Program in Cellular and Molecular Medicine. She believes the findings could also apply to other forms of autoimmune encephalomyelitis (inflammation of the brain and spinal cord), as well as inflammatory arthritis.
“We’ve demonstrated in mice you can target these cells and get rid of them,” she says. “We don’t know if this approach would be appropriate for all cases of MS, but it could be effective in the early inflammatory stages of the disease, and in targeting newly formed cells during disease exacerbations.”
Profiling MS-inducing cells
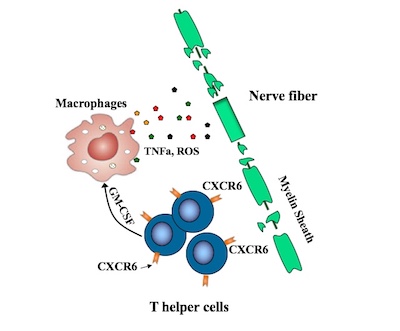
Recent efforts to pinpoint the T helper cells causing MS have pointed to TH17 cells, but some TH17 cells appear not to be involved. Remold-O’Donnell and her former postdoctoral fellow Lifei Hou, PhD, zeroed in on a subset of fast-proliferating TH17-derived cells, all bearing the CXCR6 marker. These cells, they showed, are highly damaging to nerve fibers, producing one set of proteins that directly damage cells and others, including GM-CSF, that stimulate an inflammatory attack by other immune cells known as macrophages.

The CXCR6-positive cells also produce increased amounts of a protein called SerpinB1 (Sb1), the researchers showed. When they deleted the Sb1 gene in T cells in their mouse model, fewer immune cells survived to infiltrate the spinal cord. The mice also displayed fewer symptoms of disease than control mice. Moreover, these Sb1-containing cells could be readily identified with antibodies targeting the CXCR6 surface protein.
Human counterparts
But are CXCR6+ cells relevant in human disease? To investigate, Remold-O’Donnell and Hou worked with rheumatologists, immunologists, and neuro-immunologists at Boston Children’s and Brigham and Women’s Hospital.
They obtained and tested samples of blood and synovial fluid (from the cavities of joints) from patients with inflammatory autoimmune arthritis. Levels of CXCR6+ cells were indeed elevated in the inflamed joints, but not in circulating blood from the arthritis patients, patients with MS, or healthy controls.
Targeting CXCR6 in multiple sclerosis
Remold-O’Donnell and Hou, first author on the paper, believe treatments to deplete CXCR6+ cells could mitigate MS and possibly other autoimmune disorders while largely leaving other T cell immune defenses intact. When they used monoclonal antibodies to target CXCR6, the harmful cells largely disappeared, and mice, which were primed to get MS, did not develop the disease.
The two have filed a patent covering the work and have formed a company, Edelweiss Immune, Inc., in which they have equity ownership together with Boston Children’s Hospital. The new company will be carrying the research forward.
“Many drugs have been developed to treat autoimmune diseases, such as glucocorticoids and cytotoxic reagents,” says Hou. “However, none selectively target pathogenic T cells, and long-term use of immunosuppressive agents results in broad immunosuppression and compromised immune defenses. Therapeutics with better selectivity, safety, and efficacy are needed.”
Coauthors on the study at Boston Children’s were Koichi Yuki (Anesthesiology, Critical Care and Pain Medicine), Jessica Cooley (Program in Cellular and Molecular Medicine), Lauren Henderson and Peter Nigrovic (Rheumatology), and Mark Gorman (Neurology). See the paper for a full list of authors. The work was supported by the NIH (R21 AI117440, RO1 AR065538, KO8 AR073339, K08 AR072791, P30 AR070253) and the Swiss National Science Foundation (316030_150768, 310030_146130).
More from the Program in Cellular and Molecular Medicine
Related Posts :
-

Study highlights the severity of acute necrotizing encephalopathy in kids with the flu
For most children, influenza (flu) usually means unpleasant symptoms like a fever, sore throat, and achy muscles. But for a ...
-

A world of difference: Why Noah traveled from Florida for multiple sclerosis care
When Noah started having headaches four years ago, his mom, Jonda, initially wondered if it was a classic kid ruse. “...
-

“Princess June” reigns supreme over Rasmussen syndrome
What do you call a “girly” 5-year-old who adores dolls and frilly nightgowns? If you’re one of June Pelletier’...
-

Navigating school with a neuroimmune condition
Fifteen-year-old Sarah had been challenging her dad to card games all week — and on Saturday, she finally beat him. It ...