Moving gene therapy into high gear
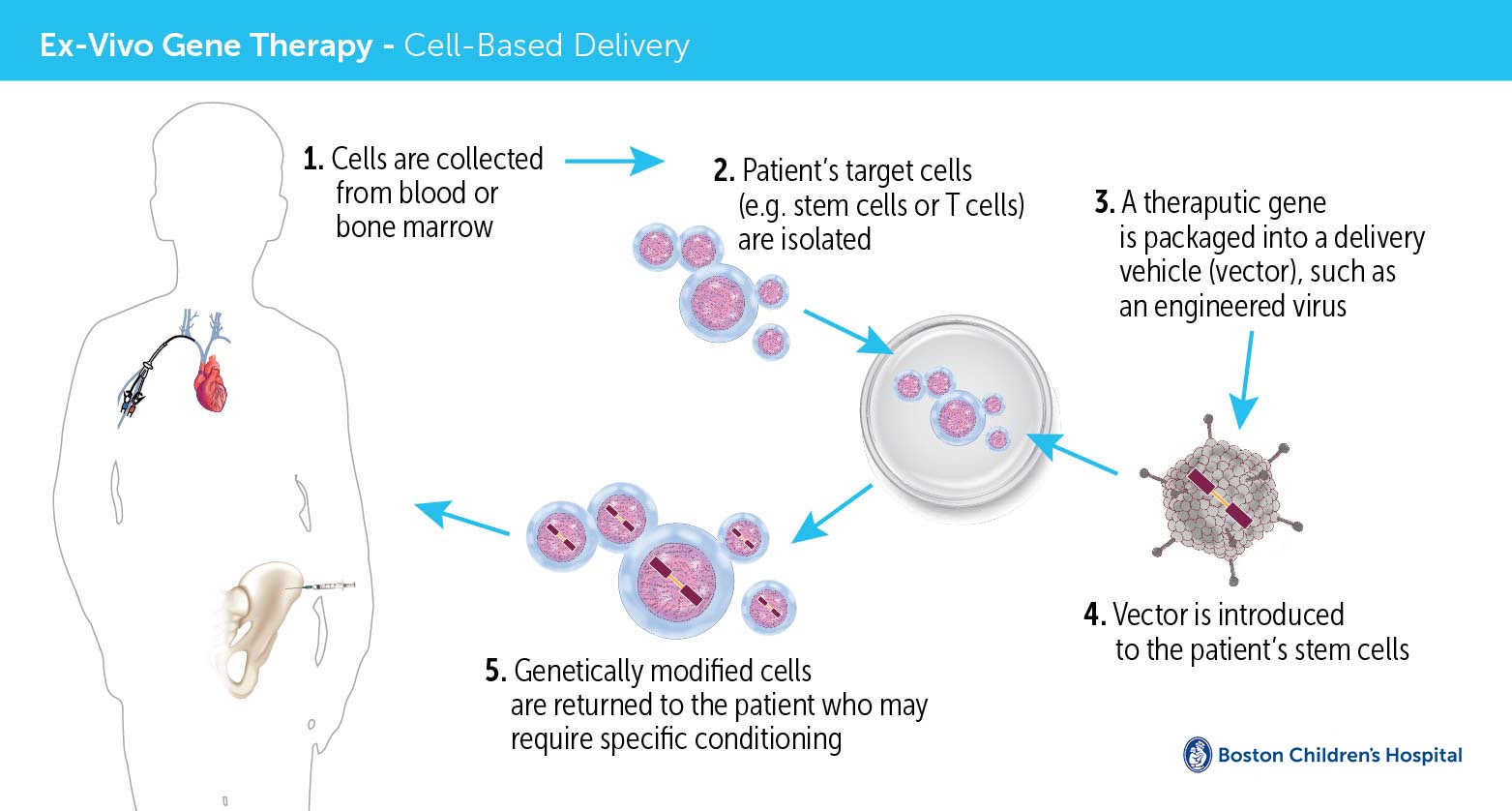
Gene therapy, still experimental but beginning to enter the clinic, attempts to utilize advanced molecular methods to treat and even reverse genetic diseases. The field started in earnest about 25 years ago and has had many setbacks along the way to its recent earliest successes.
International collaboration has been critical. Children’s Hospital Boston is one of the founding members of the Transatlantic Gene Therapy Consortium (TAGTC), a new collaboration that seeks to facilitate a more rapid advancement of this technology for treating human diseases. It was initiated shortly after the first trials of gene therapy for X-linked Severe Combined Immunodeficiency (X-SCID) (in both Paris and London) reported leukemia as a serious side effect. The TAGTC was formed to address this setback, developing safer gene therapy reagents, sharing the costs of their development, and then implementing new gene therapy trials for rare diseases across multiple international sites.
TAGTC’s initial trial in X-SCID has now opened and is accruing patients in London, Paris, Boston, Cincinnati and Los Angeles. We believe the collaborative development of this trial is a model for future international trials in rare diseases. Its development also focuses a spotlight on the ongoing challenges the field faces in implementing gene therapy trials.
Several previous single-institution trials have clearly showed the efficacy of the gene therapy approach. The Consortium targeted the critical goal of improving the safety of the vector – the virus that delivers genes into cells to correct the genetic defect (in the case of X-SCID, a defect in the blood stem cells). To compare the new vector with previous experience, the clinical protocol for the newly opened trial is essentially unchanged from the previous trials.
The Consortium members have all made significant contributions, sharing data, preclinical work, costs of vector production and, now, the treatment protocol. We believe this is a model of how rare diseases can be approached by multiple investigators, allowing us to reduce costs at each center and provide access to patients in many geographic locations.
The challenges of mounting this kind of collaborative trial are protean. Although the group adopted essentially the same trial protocol, changing only the vector, it’s taken more than two years for the trial to make its way through the regulatory and funding agencies in the United States alone. This included reviews by the National Institutes of Health (NIH) Recombinant DNA Advisory Committee (which included a full public review), the U.S. Food and Drug Administration, local Institutional Biosafety Committees (IBCs) and Institutional Review Boards (IRBs), the NIH Data and Safety Monitoring Board, the National Institute of Allergy and Infection Diseases (NIAID) and NIAID’s Division of Allergy, Immunology and Transplantation Clinical Research Committee.
A major issue we faced was that each of these reviews led to new — and often contradictory — requirements and recommendations for protocol amendments. This same process was repeated, of course, in parallel regulatory filings in the United Kingdom and in France. We were able to benefit from the TAGTC’s pooled expertise in dealing with this myriad of regulatory reviews.
While daunting in some ways, formalized scientific and clinical trials collaborations are critical to developing new therapies, in a timely and cost-effective manner, for some of the terrible genetic diseases that affect children. We are currently developing a number of other gene therapy trials using this model, to open in the next five years. Watch for trials in Wiskott-Aldrich Disease, Childhood Cerebral Adrenoleukodystrophy, Chronic Granulomatous Disease and even Sickle Cell Disease in the future.
David Williams, MD, is chief of the Division of Hematology/Oncology, co-director of the Fanconi Anemia and Bone Marrow Failure multidisciplinary clinic and director of Translational Research at Children’s Hospital Boston. A member of the NIH Recombinant DNA Advisory Committee (RAC), he has been active in development and implementation of multiple human gene therapy trials.
Visit the Gene Therapy Program
Related Posts :
-

Stay chill, show up, move forward: Nolan’s journey with ITP
At 18 years old, Nolan Ryan is an all-around athlete. Between basketball, football, and lacrosse, sports have shaped his daily life ...
-

Gene therapy for hearing loss: Tag-teaming from the lab to the clinic
Two-year-old Miles is one of the first patients with hereditary hearing loss to receive gene therapy at Boston Children’s ...
-

A true hero’s journey: How a team approach helped Wolfie overcome pancreatitis
Wolfgang, affectionately known as “Wolfie,” is a bright and energetic 7-year-old with a quick wit and a love for making ...
-

The journey to a treatment for hereditary spastic paraplegia
In 2016, Darius Ebrahimi-Fakhari, MD, PhD, then a neurology fellow at Boston Children’s Hospital, met two little girls with spasticity ...





