Lung ‘organoids’ capture early-stage lung cancer; could help test treatments
Lung cancer, the leading cancer killer in the U.S., is often missed in its earlier stages. And while recent imaging advances offer earlier detection, early-stage lung cancers still have no targeted treatments.
Key takeaways
Mini “organoids” made from lung cells offer a rapid platform for tracking early-stage lung cancers and for testing possible treatments.
RNA sequencing findings in the organoids match observations in mouse models and patient tumor samples.
But that could change. Researchers at Boston Children’s Hospital, in collaboration with Boston University and UCLA, have created an accelerated platform for identifying and testing potential treatments: “organoids” created from lung cells.
These mini lungs allowed the team, led by Carla Kim, PhD, of Boston Children’s Stem Cell Research Program, to track one common, hard-to-treat lung tumor from its origins. Studying adenocarcinoma driven by mutations in the KRAS gene, the team could capture the molecular changes that took place as it progressed. They reported their findings today in Cell Stem Cell.
Modeling early lung cancer four ways
The investigators used four parallel models of early lung cancer: tumor samples from patients with early (stage 1A) lung cancer, genetically engineered mouse models, and lung organoids. They derived the organoids either from mouse lung stem cells or from lung cells created from human induced pluripotent stem cells.
We can see changes in the organoids within seven days that can take months to see in mice and even longer, probably years, in patients.”
– Carla Kim
“We know very little about the early events that transform a normal lung epithelial cell into a cancer cell,” says Kim, co-senior author on the paper with Jane Yanagawa, MD, of UCLA and Darrell Kotton, MD, of the Boston University School of Medicine. “Here, we were able to use early-stage samples from lung cancer patients to show that our organoids truly mimic what happens in patients at the very early stages. We can see changes in the organoids within seven days that can take months to see in mice and even longer, probably years, in patients.”
Tracking lung cancer as it develops
The researchers introduced the cancer-initiating KRAS mutation into the lung organoids’ alveolar progenitor cells. They then used single-cell RNA sequencing to see what genes were turned on, or expressed, as a result.
Over time, they saw decreased expression of genes marking mature lung alveolar cells. Meanwhile, expression of genes involved in early lung development — markers of cancer progression — increased.
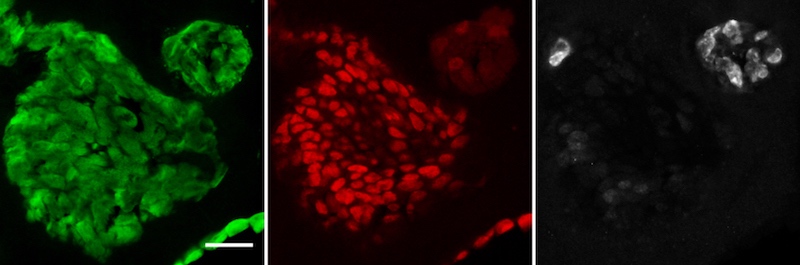
Encouragingly, studies in mice and in patient tumor samples mirrored the organoid findings.
“These studies lay a groundwork for finding new therapeutic avenues for early-stage lung cancer in the future,” says Kim. “Currently, KRAS-mutant tumors are usually resistant to therapy by the time they are diagnosed.”
Looking to the future
Although this study looked at KRAS-driven lung cancer, the researchers believe their organoid approach could facilitate the study of other kinds of cancer, including testing of candidate drugs.
“The collaborating teams really made progress together in understanding a stage of lung cancer that has been very tough to study in humans,” says Kotton. “We hope these new human and lung organoid models of early lung cancer formation will now serve as powerful drug development platforms.”
Antonella Dost and Aaron Moye, PhD, of the Boston Children’s Stem Cell Program were co-first authors on the paper.
Supporters of the study include Boehringer Ingelheim Fonds, the National Institutes of Health, the Damon Runyon Cancer Research Foundation, the Burroughs Wellcome Fund, Hope Funds for Cancer Research, an IASLC Young Investigator Fellowship, the American Cancer Society, the V Foundation for Cancer Research, the Thoracic Foundation, the Ellison Foundation, the American Lung Association, the Harvard Stem Cell Institute, and the Lung Cancer Initiative at Johnson & Johnson.
Read more from the Stem Cell Research Program and more cancer stories from Boston Children’s.
Related Posts :
-

Nanoparticle drug combo treats venous malformations
Venous malformations are abnormally shaped veins that develop when the cells lining the blood vessels grow too fast when they ...
-

Mapping ‘neighborhoods’ in aggressive childhood brain tumors
The third-most common kind of childhood brain tumor, supratentorial ependymoma, is an aggressive cancer that often returns after surgery and ...
-

A true hero’s journey: How a team approach helped Wolfie overcome pancreatitis
Wolfgang, affectionately known as “Wolfie,” is a bright and energetic 7-year-old with a quick wit and a love for making ...
-

Four things you should know about MAPCAs treatment
As the first grandchild in her family, Hannah Homan is in demand for frequent visits. She was also the focus ...





