Leading the way: Ava’s experience with teduglutide
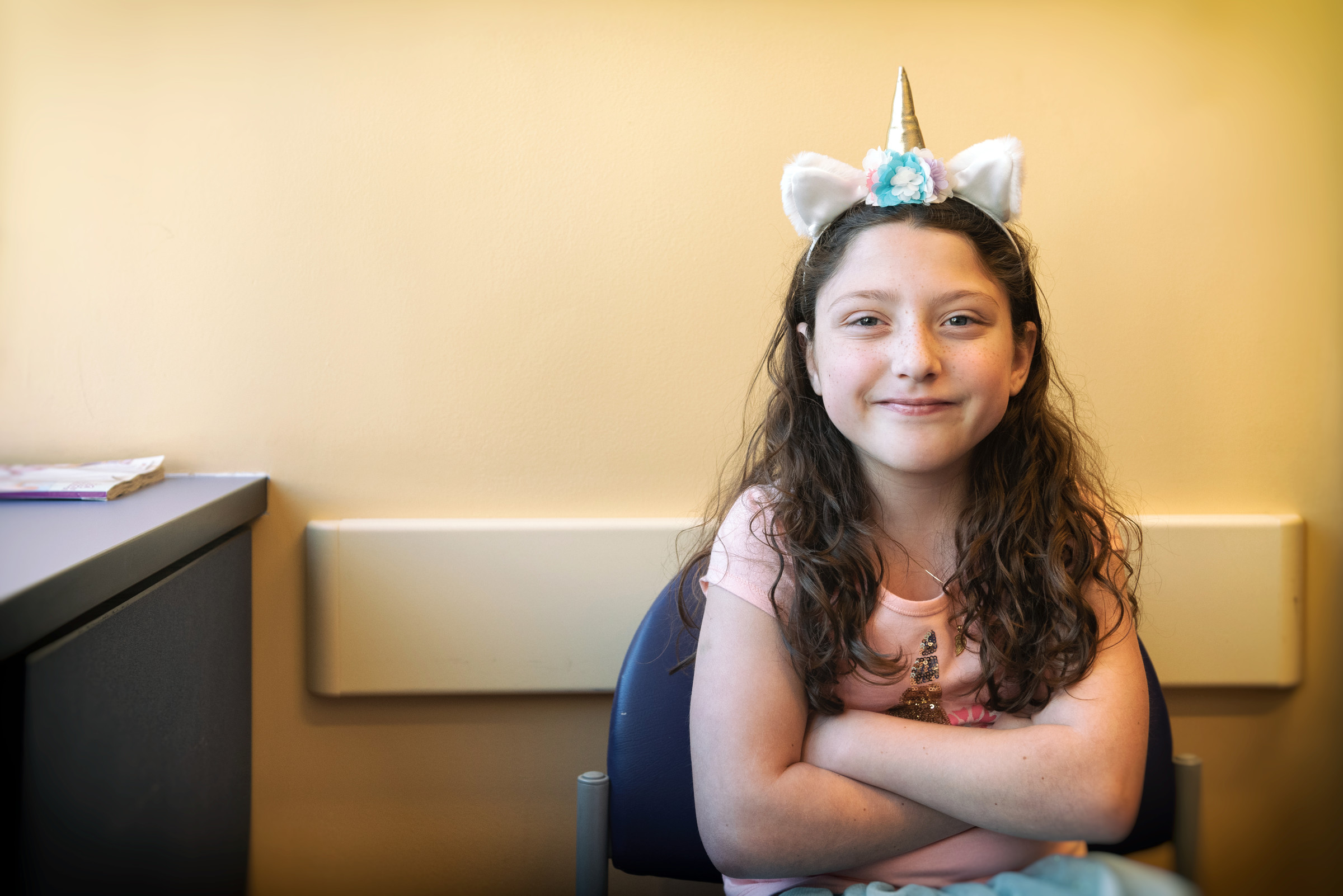
Ava Orsini visits Boston Children’s Hospital every two weeks. It’s more time than any kid wants to spend at a hospital, particularly when she lives a state away. But for this 9-year-old and her parents, the trips are worth it: For the past two years, she’s been participating in a clinical trial of a drug that has had life-changing effects on her health. Thanks to the experiences of Ava and other children, that drug, teduglutide (Gattex), was recently approved by the U.S. Food and Drug Administration (FDA) for use in eligible kids with short bowel syndrome.
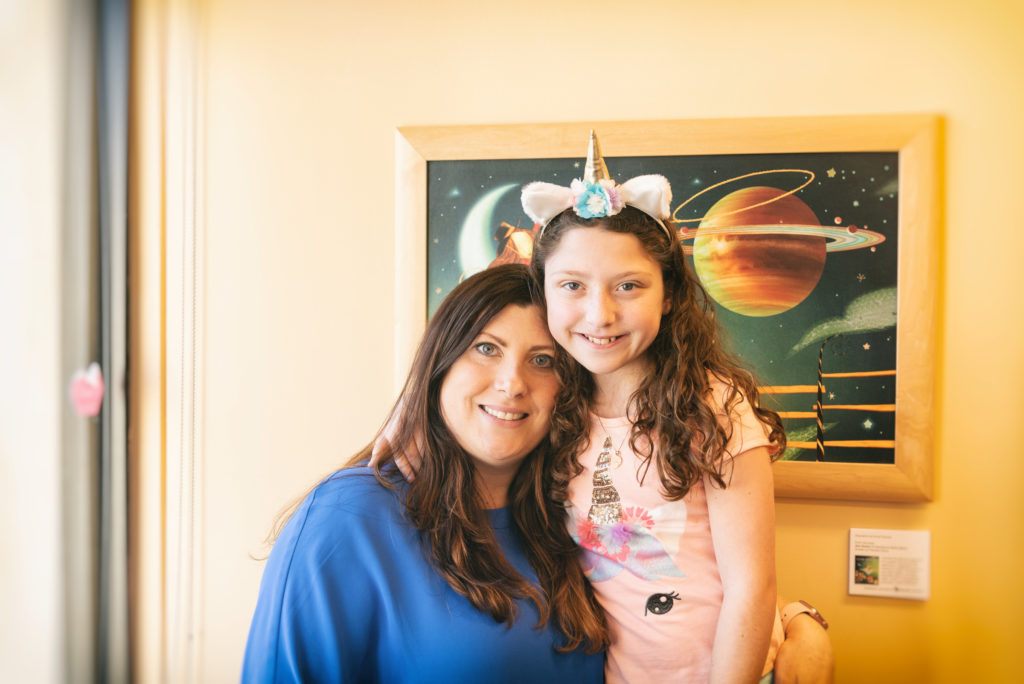
‘Almost no chance’
A twin who was born prematurely, Ava was just a week old when she developed necrotizing enterocolitis, a serious intestinal illness that leads to the death of intestinal tissue. As a result, surgeons at her local hospital in Connecticut had to remove a significant portion of her small intestine and nearly all of her large intestine. She has only has about 31 centimeters of small intestine remaining, leaving her with short bowel syndrome (SBS). In this condition, the body can’t absorb the nutrients to grow and thrive, and many children with SBS must receive nutrition intravenously through a central line (total parenteral nutrition, or TPN) or through a feeding tube.
Ava not only needed TPN, but she was also diagnosed with a congenital heart anomaly called a ventricular septal defect. “Her doctors gave her almost no chance of survival,” remembers her mother, Kate. Although surgery at 4 weeks old addressed her heart defect, Ava soon began to develop liver failure, a possible consequence of TPN.
A promising new option
Through a series of connections, Kate and Ava’s father, Bill, found Dr. Mark Puder in the Center for Advanced Intestinal Rehabilitation (CAIR) at Boston Children’s Hospital, who thought that Omegaven might help. This fish-oil based product has been shown to prevent liver damage in kids on TPN. The family traveled to Boston, where Ava began treatment with Omegaven. When she was 6 months old, she was finally able to go home, although she was still dependent on TPN for nutrition and wasn’t fully thriving.
That began to change two years ago, when Dr. Christopher Duggan, CAIR’s medical director, asked the Orsinis if they were interested in joining a study of teduglutide. Already FDA approved for use in PN-dependent adults with SBS, the injectable drug was being investigated as a potential therapy for children with the condition. Teduglutide works by helping the remaining intestine absorb more nutrients.
Not much to lose
The trial would involve daily injections of teduglutide in six-month cycles, with checkups at Boston Children’s every two weeks. “There were still a lot of unknowns with this treatment, but we decided as a family to give it a try,” says Kate. “We figured we didn’t have much to lose.”
Like most kids, Ava wasn’t thrilled with the idea of nightly shots at first. “She was petrified in the beginning,” Kate admits. But then the benefit began to become apparent. After just her first six months on teduglutide, Ava was able to wean off of TPN and onto a gastrostomy tube (G-tube). But when she entered the “washout” period of the study, during which she went off of the drug, she declined quickly. When it was time to restart teduglutide, she stayed on it.

Remarkable results
The results have been remarkable. Last November, Ava was able to have her central line removed but still uses a G-tube — for now. It’s given the whole family more freedom, and has allowed Ava to enjoy swimming without worrying about getting wet. “She can tell the difference with teduglutide,” says her mom. “She knows it’s helping her.”
When she’s not swimming, Ava loves playing tennis, doing arts and crafts, and singing with her family. “She dances to the beat of her own drum,” says Kate. And by her side through it all is Mia, her twin. “Mia gives Ava emotional support when she needs it,” Kate says. “They have a really special relationship.”
But Ava has formed another meaningful bond, too. When she first started seeing Dr. Duggan, she brought a kid-sized purse and wallet to her appointment, into which he happily dropped his business card. “He put his cell number on there and told her to call him anytime,” laughs Kate. “And one time, Ava actually did.” Today, that card has a special place in Ava’s jewelry box — and she knows that Dr. Duggan and the rest of the CAIR team will always be there when she needs them.
Learn about the Center for Advanced Intestinal Rehabilitation. Teduglutide is FDA approved for pediatric patients 1 year of age and older with SBS who require PN.
Related Posts :
-

The ‘Trach Chapter’: Isabella’s journey with bronchopulmonary dysplasia
Isabella’s life has been anything but ordinary. Born at just 27 weeks gestation and weighing only 1 pound, 4 ounces, Isabella has ...
-

Promising advances in fetal therapy for vein of Galen malformation
In 2024, Megan Ingram* of California and her husband were preparing for the birth of their third child when a 34-week ...
-

The hidden burden of solitude: How social withdrawal influences the adolescent brain
Adolescence is a period of social reorientation: a shift from a world centered on parents and family to one shaped ...
-

A toast to BRD4: How acidity changes the immune response
It started with wine. Or more precisely, a conversation about it. "My colleagues and I were talking about how some ...