When worlds collide: Glycosylated RNAs upend cell biology as we know it
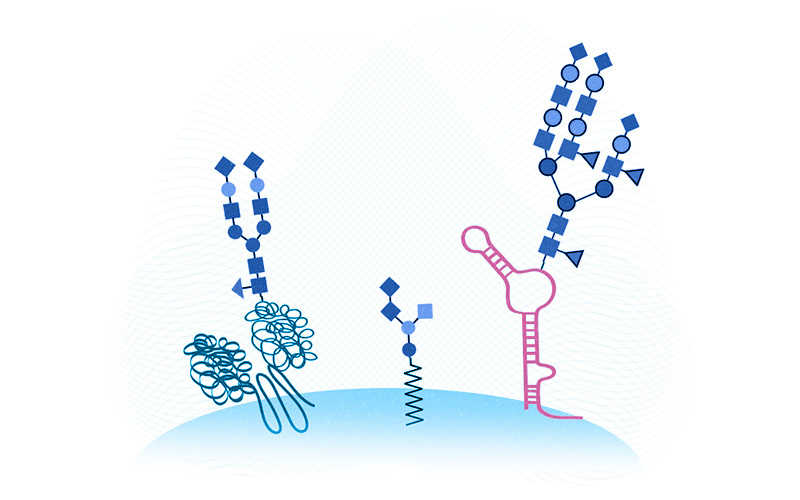
Cells in our body bristle with sugars known as glycans that other cells can recognize via specialized receptors. Glycans attach to and modify proteins and fats, thereby influencing how proteins fold, how cues are trafficked between cells, and other cell-to-cell interactions. As just one example of glycans’ importance, our blood types (A, B, O) depend on which sugars red blood cells incorporate into their protective coating.
New research, led by Ryan Flynn, MD, PhD, reveals a whole other universe of sugar-coated molecules that ride on our cells. Called glycosylated RNAs or glycoRNAs, they are surprisingly pervasive — yet nobody had previously done the right experiments to find them. And they open up another dimension for exploring how our bodies work and the origins of disease.
Flynn’s discovery of glycoRNAs, while at Stanford University in the lab of Carolyn Bertozzi, PhD, was in part serendipity. Now, in his new lab in the Stem Cell Program at Boston Children’s Hospital, he’s begun exploring these molecules’ importance. In a study just published in Cell, he and Bertozzi flesh out some early details.
“We found that glycoRNAs are on the cell surface, just like proteins and lipids,” says Flynn. “This is exciting is because it means that glycoRNAs can participate directly in cell-to-cell communication. That was previously thought to be off limits for RNAs, which had not been thought to play a role on the cell surface.”
Exploring a new biological actor
Flynn and Bertozzi revealed that these glycoRNAs consist of small, noncoding RNAs — which play known regulatory roles inside cells — coated with a class of sugars known as N-glycans. To their great surprise, glycoRNAs appear to be ubiquitous. They showed up in multiple cell types across multiple mammalian species, including humans.

But what do glycoRNAs do? And why are they important?
During their investigations, Flynn and Bertozzi noticed that some of the glycoRNAs’ N-glycans were capped by a sugar called sialic acid. This inspired them to investigate whether glycoRNAs interacted with Siglecs, certain receptors on immune cells known to bind to sialic acid.
“This was where the hair on our neck stood up,” says Bertozzi.
Certain members of the Siglec family, a class of proteins that regulate immune cells, bound cells when RNA was present. This led Flynn to suspect that glycoRNAs could help fine-tune our immune system. His new lab will investigate glycoRNAs’ potential role in autoimmune disease in collaboration with immunologists and rheumatologists at Boston Children’s.
“Ryan is starting his independent career sitting on a bombshell,” says Bertozzi.
GlycoRNA: Bridging two fields
The textbook-changing discovery of glycoRNAs was fortuitous. Flynn, who was trained in RNA biology, joined Bertozzi’s lab, which focuses on glycoscience, to better understand how glycans work. Bertozzi and others have for decades used tools from bioorthogonal chemistry, a field Bertozzi’s lab pioneered, to examine protein and lipid glycans on the cell surface.

“At the time, there was no intersection known between the RNA world and the glycan world. They were two separate areas of biology,” says Bertozzi.
Flynn became fascinated with glycans, and paired Bertozzi’s tools with methods that detect RNAs. “I joined together two spheres of information, initially with not much to go on,” he says.
Flynn began by labeling RNAs and glycans with special probes. To his surprise, the glycan probes kept turning up with RNAs — albeit very tiny RNAs, among the smallest that exist. He and Bertozzi thought at first there must be an artifact or contaminant in their samples. But Flynn confirmed the finding through a battery of experiments.
Once he proved that glycoRNAs are real, Flynn next showed that glycoRNAs are concentrated at the cell surface, just like glycoproteins and glycolipids — and ready to interact with other cells. He now wants to figure out how RNAs and glycans are able to hook up, since they traditionally inhabit different compartments of the cell.
GlycoRNAs are on the cell surface, which means they can participate directly in cell-to-cell communication. That was previously thought to be off limits for RNAs.” —Flynn
“Everyone thought we had two ‘hands’ on the cell surface, and now we know we have three,” Flynn says. “I used every single lever of RNA biology that I had. But the discovery of glycoRNAs would not have been possible had I not been in Carolyn’s lab.”
Bertozzi is a Howard Hughes Medical Institute investigator. Other supporters of the work include the National Institutes of Health, the Damon Runyon Cancer Research Foundation, the National Institute of General Medical Sciences, and the National Science Foundation. The Flynn lab is currently supported by Burroughs Welcome Fund.
Related Posts :
-

Missed signals? A new way we vary from each other biologically
Genetics has made huge strides over the past 20 years, from the sequencing of the human genome to a growing understanding ...
-

Chromatin regulation enables generation of diverse antibodies
We need a variety of antibody types to help fight off invading foreign pathogens and our genome is exquisitely tuned ...
-

Presidential awardee explores epigenetics via slime molds, worms, and more
Pretty much all of your cells have the same DNA code. Yet they come in an astonishing number of forms. ...
-

A case for Kennedy — and for rapid genomic testing in every NICU
Kennedy was born in August 2025 after what her parents, John and Diana, describe as an uneventful pregnancy. Soon after delivery, ...