Diving deep on epilepsy genetics

When child neurologist Annapurna Poduri, MD, MPH finished her clinical epilepsy fellowship at Boston Children’s Hospital in 2004, she was struck to find that the genetic understanding of epilepsy had changed little in the decade since she started medical school. Many questions were unanswered — and some weren’t yet being asked. Existing treatments were still not working in a third of patients. Some needed brain surgery for relief from seizures.
Seeing the devastating effects of epilepsy on patients and families, Poduri felt it was urgent to move the genetic knowledge base forward. Drawing insight from her patients at Boston Children’s, and by studying their brain tissue directly, she sought to establish how somatic mutations, arising during early brain development, could lead to epilepsy. With a team of physicians, genetic counselors, researchers, and students, she began to identify epilepsy genes. In 2011, she launched the Epilepsy Genetics Program clinic and eventually a laboratory, advancing care one child at a time.
Building a portfolio of epilepsy genes
To date, Poduri and her colleagues have identified dozens of genetic causes of epilepsy. Some of these are exquisitely subtle. One of the first was found in a boy with daily seizures whose entire right brain was malformed and enlarged; the genetic abnormality was detectable in only about a third of his brain cells. Other patients have had even smaller percentages of brain cells affected, requiring deep genomic sequencing to identify their mutations.
A SEIZURE SUCCESS STORY
Pierce and Albry, siblings with severe epilepsy, were able to receive a custom treatment based on their genetic profile. Pathogenic variants in a gene called CAD prevented them from synthesizing uridine, a component of nucleic acids especially needed in the developing brain. With this genetic diagnosis, they were treated with readily available uridine supplements. Albry, whose treatment began at age 5, had dramatic improvements in seizure control; Pierce, age 14, saw his condition stabilize.
As genomic sequencing has become more routine, discovery has been accelerated. Through the hospital-wide Children’s Rare Disease Cohorts initiative, Poduri’s team has now sequenced the genomes of more than 500 patients. “We’ve gotten genetic answers for over 100 children, which is amazing,” she says.
As genetic variants are discovered in children with epilepsy, each is carefully investigated. Through literature searches, consults with other experts, cell models, and sometimes animal models, researchers can confirm whether a variant is truly causative, explore how it causes epilepsy, and most important, determine whether its effects could be reversed (see sidebar). Genes making active protein in the brain and those that seem to be involved in neuronal excitability go to the top of the list for further vetting.
Fishing for epilepsy treatments
The Epilepsy Genetics team can now model patients’ genes and test treatments at growing scale thanks to a new research partner: a tiny aquarium fish. Whereas mice are slightly closer to humans genetically, zebrafish have more than 80 percent of the same genes we do. And when genes involved in human epilepsy are perturbed, they sometimes show evidence of seizure activity.

Early on, Poduri teamed up with fellow neurologist Alexander Rotenberg, MD, PhD, who developed an ultra-miniaturized EEG apparatus for zebrafish. With it, they can detect whether fish with introduced mutations in epilepsy genes are more seizure-prone than their companions.
“Zebrafish grow fast, so within five or six days you can look for core features of epilepsy, like hyperexcitability and fast swirling movements that approximate seizure-like behaviors,” says Poduri. “Of our 100 newest candidate genes, we have a short list of 12 that we will start to investigate. By the end of the year, if we’re lucky, we’ll have three or four that cause seizure activity in zebrafish and will be able to test seizure medications.”
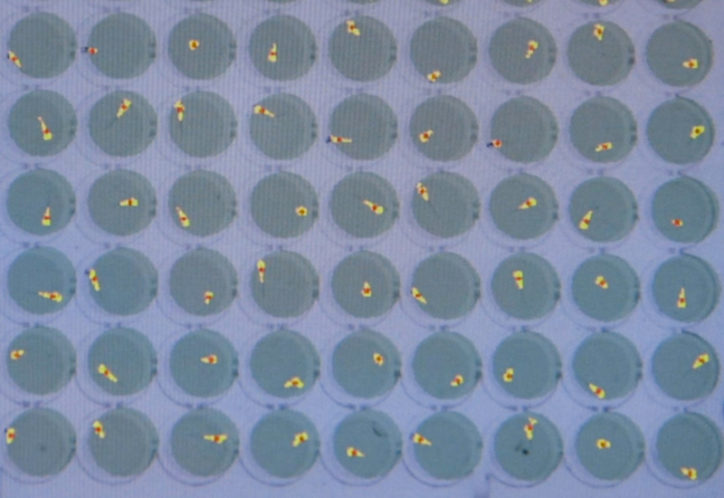
The tiny size of zebrafish embryos allows for rapid, scaled-up drug testing. This involves loading the fish into 96-well plates and adding different medications — from a library of known drugs — into the fishes’ water. Automated detectors can then capture the fishes’ movements and brain activity and flag those whose seizures disappear when a given drug is added.
Advancing epilepsy care through genetics
Based on Poduri’s work, some patients have had their epilepsy medications adjusted, and some are receiving gene-specific treatments. But ultimately, Poduri’s goal is to transform the entire approach to diagnosing and treating patients with epilepsy. Through a global genetics collaboration known as the International Precision Child Health Partnership (IPCHiP), she and her colleagues are leading a precision medicine project to determine the potential of genetics to change routine care and patient outcomes.
An early report from the team indicates that genetic testing benefits children with unexplained epilepsy. In a cohort of 152 patients, 72 percent had changes to their medical management based on the results of genetic testing.
“We hope to demonstrate the impact of early genetic diagnosis in babies with epilepsy,” says Poduri. “When they come to the hospital with their first seizure, we want to diagnose them quickly and find ways to change their treatment based on their genetics. And eventually maybe even diagnose infants with a tendency for epilepsy before they have seizures. We’re collaborating across labs, across departments, across institutions, and across countries to be able to move things faster.”
Visit the Epilepsy Genetics Program and the Poduri Lab.
Related Posts :
-

A promising new antiseizure drug tailored to newborns
Neonatal seizures can lead to serious consequences, including significant cognitive and motor disabilities, lifelong epilepsy, and death. They are ...
-

After decades of evolution, gene therapy arrives
As early as the 1960s, scientists speculated that DNA sequences could be introduced into patients’ cells to cure genetic disorders. ...
-

Mary’s journey with genetic epilepsy
Lots of people don’t like the heat. It makes them feel uncomfortable and sluggish. But for Mary, being outside ...
-

I tried the keto diet to help my epilepsy and it changed my life
My name is Dennis. I’m 15 years old and live in East Greenwich, Rhode Island. Two years ago, I started ...





