Beth Stevens: A transformative thinker in neuroscience
When 2015 MacArthur “genius” grant winner Beth Stevens, PhD, began studying the role of glia in the brain in the 1990s, these cells—“glue” from the Greek—weren’t given much thought. Traditionally, glia were thought to merely protect and support neurons, the brain’s real players.
But Stevens, from the Department of Neurology and the F.M. Kirby Neurobiology Center at Boston Children’s Hospital, has made the case that glia are key actors in the brain, not just caretakers. Her work—at the interface between the nervous and immune systems—is helping transform how neurologic disorders like autism, amyotrophic lateral sclerosis (ALS), Alzheimer’s disease and schizophrenia are viewed.
Soon after college graduation in 1993, without prior experience in neuroscience, she helped discover an interplay between neurons and glial cells known as Schwann cells that controlled production of the nerve insulation known as myelin It was one of the early pieces of evidence that glia and neurons talk to each other.
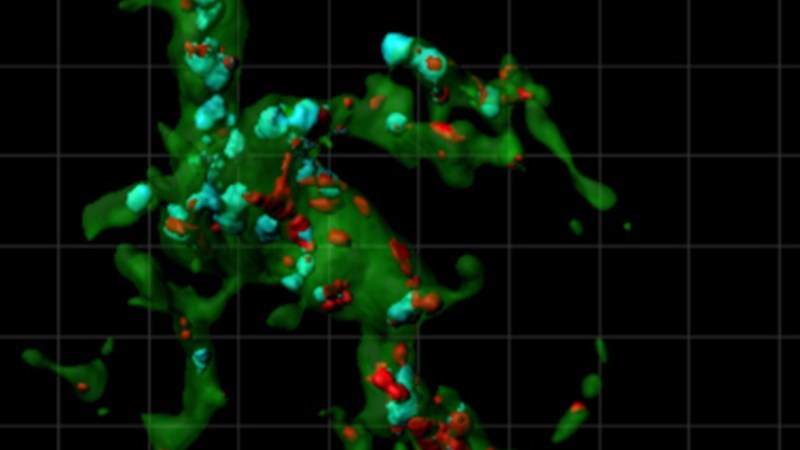
In 2007, while still a postdoctoral fellow, Stevens showed how star-shaped glial cells called astrocytes influence the development of synapses, or brain connections. Studying neurons, her lab showed that a gene called C1q was markedly more active when astrocytes were present. C1q is an immune gene, one nobody had expected to see in a normal brain. In the context of disease, it initiates the complement cascade, an immunologic pathway for tagging unwanted cells and debris for clearance by other immune cells.
But in healthy developing brains, Stevens showed, C1q was concentrated at developing synapses, or brain connections, apparently marking certain synapses for pruning.
Then in 2012, the Stevens lab showed that microglia—another type of glia usually thought of as immune cells themselves—actively sculpt the brain’s wiring. They literally trim away unwanted, inappropriate synapses by eating them—in the same way they’d engulf and destroy invading bacteria.
That paper was cited by the journal Neuron as the year’s most influential paper.
The same year, she received a Presidential Early Career Award for Scientists and Engineers, honoring her innovative research and scientific leadership.
Stevens’s current investigations are looking at synapse loss—a hallmark of neurodegenerative conditions such as Alzheimer’s—and trying to understand why it occurs. Her lab’s recent work suggests that normal pruning mechanisms that are active during early brain development get re-activated later in life. Intervening with this activation could lead to a new treatment approach, she believes.
Stevens isn’t the only brain researcher at Boston Children’s to become a MacArthur fellow. Neurosurgeon Benjamin Warf, MD, received the honor in 2012.
Related Posts :
-

The hidden burden of solitude: How social withdrawal influences the adolescent brain
Adolescence is a period of social reorientation: a shift from a world centered on parents and family to one shaped ...
-

The journey to a treatment for hereditary spastic paraplegia
In 2016, Darius Ebrahimi-Fakhari, MD, PhD, then a neurology fellow at Boston Children’s Hospital, met two little girls with spasticity ...
-

The thalamus: A potential therapeutic target for neurodevelopmental disorders
Years ago, as a neurology resident, Chinfei Chen, MD, PhD, cared for a 20-year-old woman who had experienced a very ...
-

Could peripheral neuropathy be stopped before it starts?
An increase in high-fat, high-fructose foods in people’s diets has contributed to a dramatic increase in type 2 diabetes. This, ...