Inhibiting inhibitory neurons gets mice with spinal cord injury to walk again
Most people with spinal cord injury are paralyzed from the injury site down, even when the cord isn’t completely severed. Why don’t the spared portions of the spinal cord keep working, allowing at least some movement? A new study just published online by Cell provides insight into why these nerve pathways remain quiet. Most intriguingly, it shows that injection with a small-molecule compound can revive these circuits in paralyzed mice — and get them walking again.
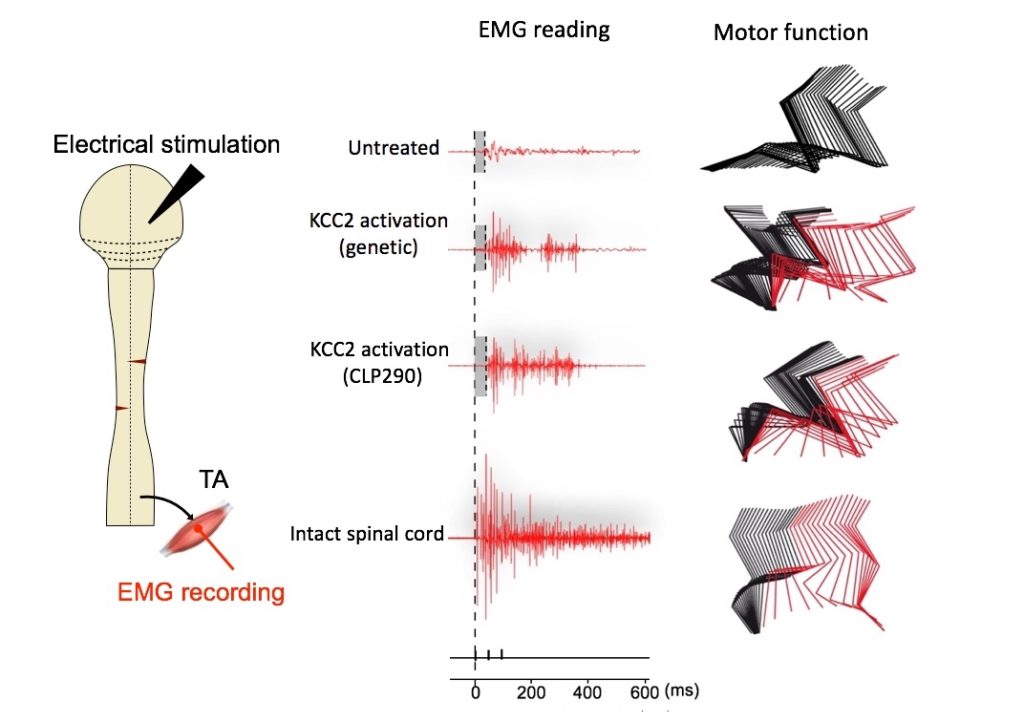
“We saw 80 percent of mice treated with this compound recover their stepping ability,” says Zhigang He, PhD, of Boston Children’s Hospital’s F.M. Kirby Neurobiology Center, the study’s senior investigator. “For this fairly severe type of spinal cord injury, this is the most significant functional recovery we know of.”
Taking a cue from the clinic
Many animal studies looking to repair spinal cord damage have focused on getting nerve fibers, or axons, to regenerate, or getting new axons to sprout from healthy ones. While He’s lab and others have achieved impressive regeneration and sprouting, the corresponding effects on motor function after a severe injury are less clear. Some studies have tried using neuromodulators such as serotonergic drugs to simulate the spinal circuits, but have gotten only transient, uncontrolled limb movement.
The new study took a different approach, which could complement these other strategies. The researchers had noted that epidural electrical stimulation, which applies a current to the lower portion of the spinal cord, has enabled some patients to regain movement when combined with rehabilitation training. To date, it’s the only clinical treatment that’s been found effective.
“Epidural stimulation seems to affect the excitability of neurons,” says He. “However, in these studies, when you turn off the stimulation, the effect is gone. We tried to come up with a pharmacologic approach to mimic the stimulation and better understand how it works.”
Altering excitability
He, first author Bo Chen and colleagues selected a handful of compounds that are already known to alter the excitability of neurons, and are able to cross the blood-brain barrier. They gave each compound to paralyzed mice in groups of 10 via intraperitoneal injection. All mice had severe spinal cord injury, but with some nerves intact. Each group (plus a control group given placebo) was treated for eight to ten weeks.
One compound, CLP290, had the most potent effect, enabling paralyzed mice to regain stepping ability after four to five weeks of treatment. Electromyography recordings showed that the two relevant groups of hindlimb muscles were active. The animals’ walking scores remained higher than the controls’ up to two weeks after stopping treatment. Side effects were minimal.
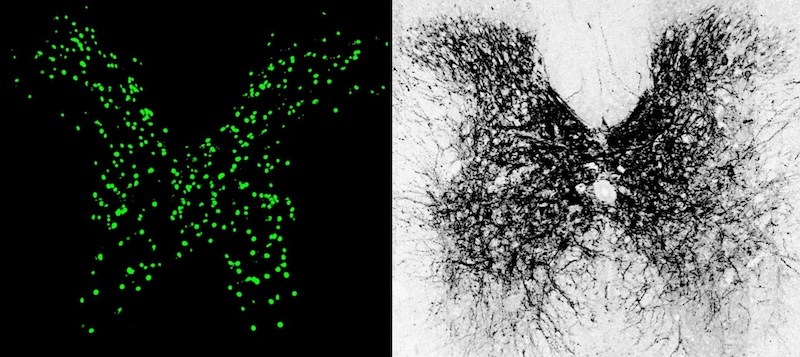
Restoring inhibition to increase excitation
The research findings show that inhibitory neurons in the injured spinal cord are crucial to recovery of motor function. CLP290 is known to activate a protein called KCC2, found in cell membranes, that transports chloride out of neurons.
After spinal cord injury, inhibitory neurons produce dramatically less KCC2. As a result, He and colleagues found, they can’t properly respond to inhibitory signals from the brain. Instead, they respond only to excitatory signals that tell them to keep firing. And since these neurons’ own signals are inhibitory, the result is too much inhibitory signaling in the local spinal circuit. Bottom line: The brain’s commands telling the limbs to move aren’t relayed.
Restoring inhibition will allow the whole system to be excited more easily.
By restoring KCC2, with either CLP290 or genetic techniques, the inhibitory neurons can again process inhibitory signals from the brain, so they fire less. This shifts the overall circuit back toward excitation, the researchers found, making it more responsive to input from the brain. This had the effect of reanimating spinal circuits disabled by the injury.
“Restoring inhibition will allow the whole system to be excited more easily,” He explains. “Too much excitation not good, and too much inhibition is not good either. You really need to get a balance. This hasn’t been demonstrated in a rigorous way in spinal cord injury before.”
He and colleagues are now investigating other compounds that act as KCC2 agonists. They believe such drugs could be combined with epidural stimulation to maximize a patient’s function after spinal cord injury. Gene therapy to restore KCC2 is another possibility.
“We are very excited by this direction,” says He. “We want to test this kind of treatment in a more clinically relevant model of spinal cord injury and better understand how KCC2 agonists work.”
Bo Chen, Yi Li (Boston Children’s Hospital) and Bin Yu (Nantong University, China) were co-first authors on the paper. Xiosong Gu (Nantong University) and Zhigang He are co-senior authors. Coauthors were Zicong Zhang, Benedikt Brommer, Philip Raymond Williams, Yuanyuan Liu, Shane Vincent Hegarty, Junjie Zhu and Yiming Zhang (Boston Children’s Hospital); Songlin Zhou (Nantong University); Hong Guo and Yi Lu (Brigham and Women’s Hospital, Boston).
The study was supported by the National Major Project of Research and Development of China (2017YFA0104701), the National Institute of Neurological Disorders and Stroke (NS096294), the Craig Neilsen Foundation, the Paralyzed Veterans of America Research Foundation and the Dr. Miriam and Sheldon G. Adelson Medical Research Foundation.
Related Posts :
-

The hidden burden of solitude: How social withdrawal influences the adolescent brain
Adolescence is a period of social reorientation: a shift from a world centered on parents and family to one shaped ...
-

The journey to a treatment for hereditary spastic paraplegia
In 2016, Darius Ebrahimi-Fakhari, MD, PhD, then a neurology fellow at Boston Children’s Hospital, met two little girls with spasticity ...
-

Parsing the promise of inosine for neurogenic bladder
Spinal cord damage — whether from traumatic injury or conditions such as spina bifida — can have a profound impact on bladder ...
-

The thalamus: A potential therapeutic target for neurodevelopmental disorders
Years ago, as a neurology resident, Chinfei Chen, MD, PhD, cared for a 20-year-old woman who had experienced a very ...





