Inherited autism mutations found via genomic sequencing in Mideast families
Autism clearly runs in some families, yet few inherited genetic causes have been found. A major reason is that these causes are so varied that it’s hard to find enough people with a given mutation to establish a clear pattern. Now, three large Middle Eastern families with autism spectrum disorders (ASDs) have led the way to a few more mutations, potentially broadening the number of genetic tests available to families.
What’s fascinating is that the mutations, described earlier this week in Neuron, affect genes known to cause severe, often lethal genetic syndromes. Milder mutations in the same genes, found through genomic sequencing, primarily cause autism.

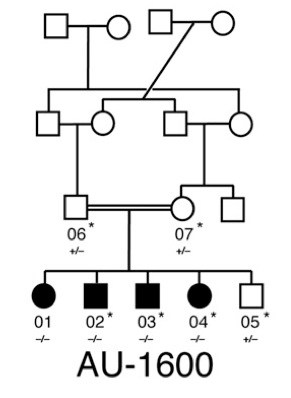
Researchers Tim Yu, MD, PhD, Maria Chahrour, PhD, and senior investigator Christopher Walsh, MD, PhD, of Boston Children’s Hospital, started with three large families that had two or more children with an ASD, in which the parents were first cousins. Cousin marriages are a common tradition in the Middle East that greatly facilitates the identification of inherited mutations—as does large family size.
“Families from the U.S. are not ideal for finding inherited genetic mutations, since family sizes are often small,” says Walsh, chief of Genetics at Boston Children’s and an investigator of the Howard Hughes Medical Institute.
The researchers first used genetic mapping techniques to narrow their search to chromosome regions with recessive mutations, requiring a double hit to cause disease. They then sequenced the protein-coding genes in those areas—otherwise known as whole-exome sequencing. That turned up mutations in three genes not previously known to be involved in autism:
- AMT, a gene classically associated with nonketotic hyperglycinemia, a severe metabolic syndrome marked by severe seizures and death during infancy.
- PEX7, classically associated with rhizomelic chondrodysplasia punctata, a severe syndrome causing metabolic and bone abnormalities, cataracts, severe epilepsy and early death.
- SYNE1, a gene associated with brain malformation, severe motor and muscle problems, and possibly bipolar psychiatric disease.
“We have textbook descriptions of all these diseases, but in real life, there can be atypical, milder presentations.”
The severe syndromes linked to these genes do often include autistic behavior or intellectual disability, but not as the primary symptom. These milder mutations seem to cause disease that is more brain-specific.
“The AMT and PEX7 mutations weren’t picked up by standard tests for metabolic disorders,” notes Chahrour. “But when you’re able to sequence the entire exome, you can find them.”
Intrigued, the team broadened its inquiry—sequencing 70 other genes associated with genetic syndromes affecting cognition and behavior in 163 Middle Eastern families. That turned up even more interesting leads, including:
- An additional family with a recessive mutation in AMT
- Two families with recessive mutations in VPS13B, a gene known to cause Cohen syndrome, which includes intellectual disability, obesity, vision and joint problems, and small head size
- A family with a recessive mutation in POMGNT1, a gene known to cause muscle-eye-brain disease, marked by brain malformation, intellectual disability, muscle and vision problems
- A family with an X-linked mutation in MECP2 in two boys (MECP2 mutations are known to cause Rett syndrome in girls, but are typically lethal in boys).
“We have textbook descriptions of all these diseases, but in real life, there can be atypical, milder presentations,” says Yu. “The kids we were studying with autism were alive at age 13. They had double hits for these mutations, but they were much milder mutations. The proteins retained a bit of their function.”
The team also examined a cohort of U.S. patients, looking for recessive mutations in six of the genes they identified. They analyzed whole-exome sequence data from 612 families with ASDs, part of a registry known as the Simons Simplex Collection. The analysis found evidence for causative recessive mutations in at least two of the genes identified in the Middle Eastern families, suggesting that broadening the search to examine all 70 genes would be worthwhile.
“It’s not clear yet how many U.S. families have these recessive mutations,” says Yu. “Further studies could begin to estimate what fraction of autism cases might fall under this model.”
The Boston Children’s study complements another study in the same issue of Neuron, led by Mark Daly, PhD, of Massachusetts General Hospital and the Broad Institute. That study looked for recessive mutations across the entire genome in 933 cases and 869 controls—but took a different approach, specifically seeking mutations that completely knocked a gene out or abolished its function.
“Together, these two studies firmly establish that recessive mutations contribute importantly to autism, not just in specialized populations but in the population at large,” says Yu. “Genome sequencing is going to be a huge advance in identifying more of these mutations, since there are a lot of rare syndromes that are otherwise very difficult to detect.”
Related Posts :
-

A case for Kennedy — and for rapid genomic testing in every NICU
Kennedy was born in August 2025 after what her parents, John and Diana, describe as an uneventful pregnancy. Soon after delivery, ...
-

The journey to a treatment for hereditary spastic paraplegia
In 2016, Darius Ebrahimi-Fakhari, MD, PhD, then a neurology fellow at Boston Children’s Hospital, met two little girls with spasticity ...
-

New research paves the way to a better understanding of telomeres
Much the way the caps on the ends of a shoelace prevent it from fraying, telomeres — regions of repetitive DNA ...
-

New research sheds light on the genetic roots of amblyopia
For decades, amblyopia has been considered a disorder primarily caused by abnormal visual experiences early in life. But new research ...