Disulfiram inhibits inflammatory gatekeeper protein: Could it be helpful in COVID-19?
Inflammation is the alarm system by which cells first respond to potential danger. But in excess, inflammation can be deadly. In new research from the Program in Cellular and Molecular Medicine (PCCM) at Boston Children’s Hospital, scientists discovered that disulfiram, an FDA-approved drug commonly used for treating alcoholism, blocks a key gatekeeper protein involved in inflammation.
Activation of that protein, gasdermin D, is the final common step in the process of inflammatory cell death, or pyroptosis, and the resulting release of inflammatory cytokines seen in many serious conditions including sepsis. In mouse studies, animals treated with disulfiram did not develop fatal sepsis compared with untreated animals. These findings were published in a paper in Nature Immunology.

“This research discovery is coincidentally very timely today because most people think that the clinical deterioration of COVID-19 patients is mediated by a cytokine storm, or excessive release of inflammatory molecules,” explains Judy Lieberman, MD, PhD, co-senior investigator of the study together with Hao Wu, PhD, also in the PCMM and professor of structural biology at Harvard Medical School.

“Even though there has been a lot of interest, there have not been any bona fide gasdermin D inhibitors,” says Wu. “We screened thousands of compounds and found that the one that worked best — disulfiram — is already on the market, is inexpensive, has a 70-year history of drug safety, and could be repurposed pretty quickly.”
The cascade to cell inflammation
When an invading virus or bacteria enter a cell, it triggers inflammation unleashing a cascade of events. One key event is called pyroptosis, or a fiery or inflammatory cell death. In pyroptosis, the cell’s membrane literally explodes, releasing inflammatory molecules like interleukin-1, which causes fever.
In a paper published in Nature in 2016, Lieberman and Wu discovered that gasdermin D forms membrane pores. When these pores open, inflammatory molecules spill out of the cell causing pyroptosis.
Too much inflammation contributes to human diseases, including sepsis, inflammatory bowel disease, gout, type II diabetes, cardiovascular disease, Alzheimer’s disease, and is responsible for rare inflammatory genetic diseases.
“We knew that gasdermin D is the gatekeeper of the pathway leading to pyroptosis and spillage of inflammatory cytokines,” says Wu. “If we could find a compound that would inhibit this particular step, that could be an attractive drug target to prevent pyroptosis when it was not needed.”
Disulfiram quiets pyroptosis
Jun Jacob Hu, PhD, screened more than 3,700 small molecules in the Wu lab looking for gasdermin D inhibitors. He found just 22 active compounds and disulfiram was at the top of the list.
Next, the team studied mice sick with sepsis. They observed that disulfiram blocked pyroptosis and its explosive release of inflammatory molecules. Mice treated with disulfiram survived while those not receiving the drug died from sepsis within one day.
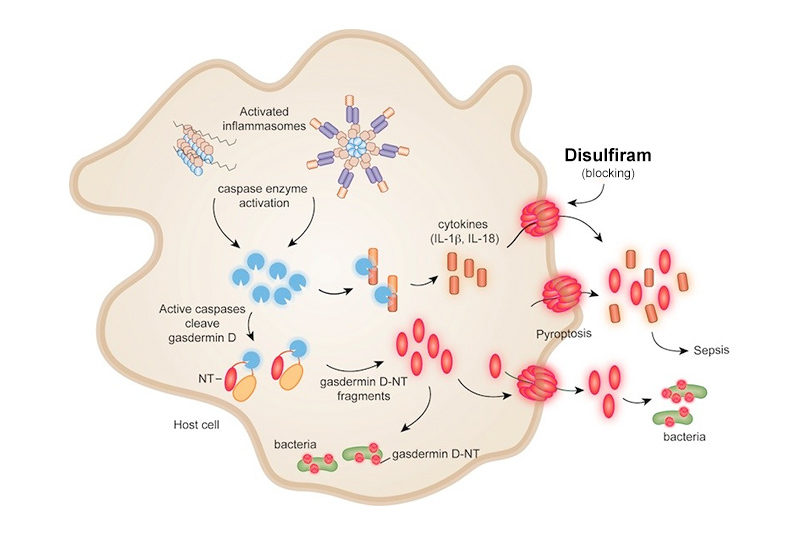
“There have been hundreds of clinical trials looking for drugs to stop sepsis and the overwhelming inflammatory response without success,” says Lieberman. Sepsis is the leading cause of death in children in the world and contributes to about a third of deaths in hospitalized adults.
“We hope that with this new discovery — inhibiting gasdermin D that is in a critical location in the inflammatory pathway — we could actually have a therapy that might work,” adds Lieberman.
COVID-19 studies planned
The team is now looking to apply these findings to COVID-19.
“Because COVID-19 can produce an inflammatory syndrome that is very similar to sepsis, we wonder whether disulfiram can be used to treat severely ill COVID-19 patients,” says Wu. “We know from a recent report that disulfiram also inhibits a coronavirus protease, one of the essential proteins of the virus causing COVID-19.”
Plans are already in place to study pyroptosis and coronavirus. The ultimate goal is to start a clinical trial with disulfiram in COVID-19 patients.
“The truth is that gasdermin D contributes to so much pathology in the body that we think an effective gasdermin D inhibitor like disulfiram could open up many therapeutic possibilities,” says Lieberman.
Additional authors of the study are Jun Jacob Hu, Xing Liu, Shiyu Xia, Zhibin Zhang, Ying Zhang, Jingxia Zhao, Jianbin Ruan, Xuemei Luo, Yang Bai, Junhong Wang, L. Robert Hollingsworth, Venkat Giri Magupalli, Li Zhao, Hongbo R. Luo, and Justin Kim.
This study was supported in part by the US National Institutes of Health (grants DP1HD087988, R01Al139914, R01AI123265, R01AI142642, R01AI145274, R01AI141386, R01HL092020 and P01HL095489), National Natural Science Foundation of China (grant 31972897), Chinese Academy of Sciences (grant ZDBS-LY-SM008), Shanghai Municipal Science and Technology (grant no. 2019SHZDZX02) Shanghai Science and Technology Committee (grant 19QA1409800) and Flight Attendant Medical Research Institute (CIA123008).
Learn more about Boston Children’s response to COVID-19 and coronavirus research at the hospital.
Related Posts :
-

A surprising link between Crohn’s disease and the Epstein-Barr virus
Crohn’s disease, a debilitating inflammatory bowel disease, has many known contributing factors, including bacterial changes in the microbiome that ...
-

Could peripheral neuropathy be stopped before it starts?
An increase in high-fat, high-fructose foods in people’s diets has contributed to a dramatic increase in type 2 diabetes. This, ...
-

Could we cure or prevent food allergy by targeting an intestinal protein?
When is food simply nourishing and enjoyable, and when does it provoke an allergic reaction? The answer appears to lie ...
-

Model enables study of age-specific responses to COVID mRNA vaccines in a dish
mRNA vaccines clearly saved lives during the COVID-19 pandemic, but several studies suggest that older people had a somewhat reduced ...